1 Department of Biosystems Technology, Faculty of Technology, University of Sri Jayewardenepura, 10206 Homagama, Sri Lanka
2 Department of Materials and Mechanical Technology, Faculty of Technology, University of Sri Jayewardenepura, 10206 Homagama, Sri Lanka
3 Cardiometabolic and Endocrine Institute, North Brunswick, NJ 08902, USA
Abstract
Human skin is a physical and biochemical barrier that protects the internal body from the external environment. Throughout a person’s life, the skin undergoes both intrinsic and extrinsic aging, leading to microscopic and macroscopic changes in its morphology. In addition, the repair processes slow with aging, making the older population more susceptible to skin diseases. Intrinsic factors associated with advanced age gradually degrade the dermal collagen matrix, resulting in fine wrinkles and reduced elasticity; this is accelerated in post-menopausal women due to estrogen deficiency. In contrast, extrinsic factors associated with advanced age, primarily caused by exposure to ultraviolet (UV) radiation, lead to coarse wrinkles, solar elastosis, hyperkeratosis, irregular pigmentation, and skin cancers. UVB radiation, while contributing to skin photo-aging, also induces the cutaneous synthesis of vitamin D. Vitamin D, in turn, protects the skin from oxidative stress, inflammation, and DNA damage, thereby delaying both chronological and photo-aging. Moreover, research has demonstrated an association between lower vitamin D levels and a higher prevalence of certain cutaneous diseases. This review explores and summarizes the critical role of vitamin D in skin aging and age-related skin diseases. The data presented highlight the importance of maintaining vitamin D adequacy throughout life.
Keywords
- vitamin D
- skin aging
- age-related skin diseases
The skin, the largest organ in the human body, acts as a physical barrier that safeguards internal structures from external threats. The skin comprises three layers: the epidermis, dermis, and hypodermis, with each layer exhibiting distinct anatomical features. The outermost layer, the epidermis, protects the skin from external damage and maintains hydration. It primarily consists of keratinocytes originating from the basal layer and moving upward toward the surface (Fig. 1).
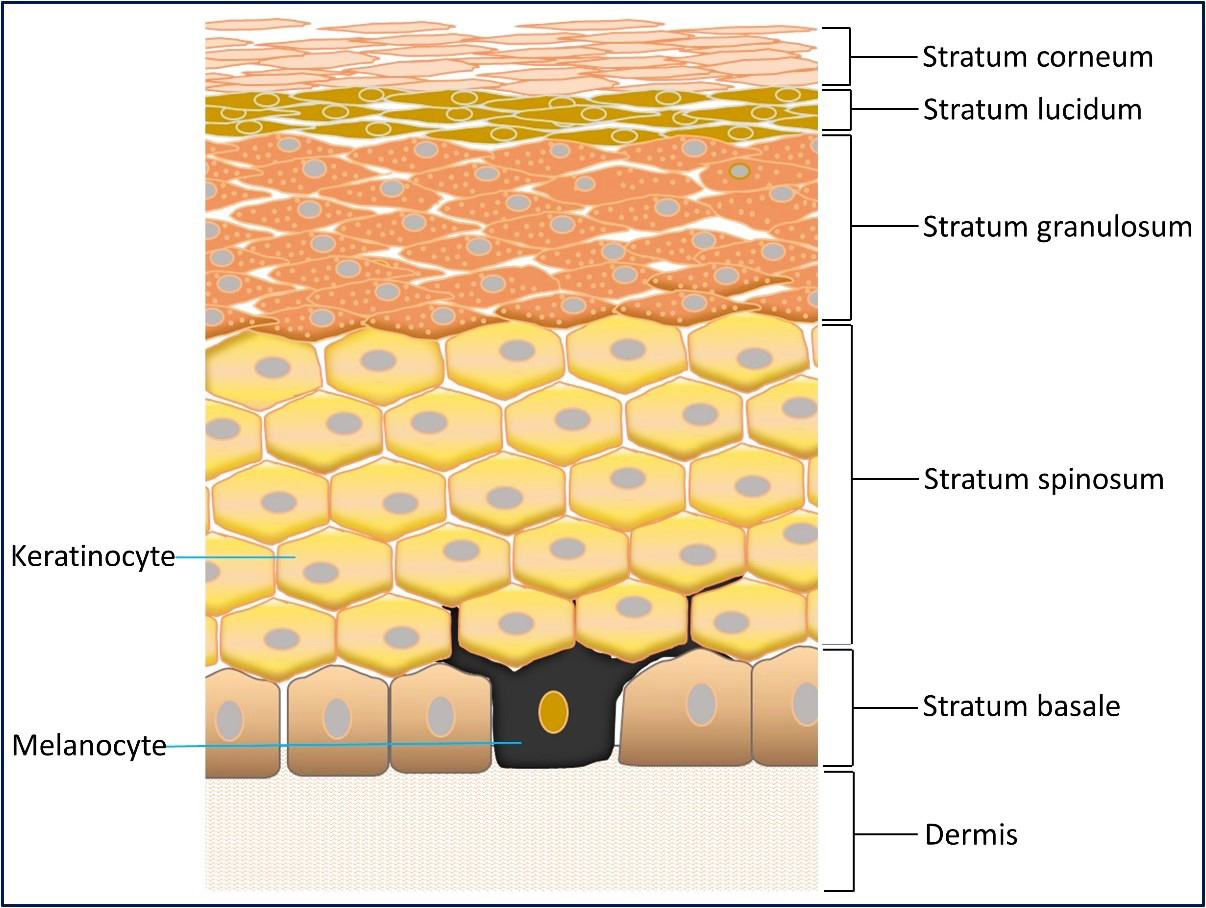 Fig. 1.
Fig. 1.
The multilayer structure of the epidermis. The image shows multiple layers of the epidermis, the keratinocytes and melanocytes, and part of the dermis.
As keratinocytes ascend, they become highly keratinized, mature, change shape, and join the stratum corneum, the dead surface layer of the skin [1, 2, 3]. The epidermis also includes melanocytes, Langerhans cells, and Merkel cells. Melanocytes produce melanin, a pigment that gives the skin its color. Antigen-presenting dendritic Langerhans cells act as the skin’s first line of defense, and Merkel cells in the stratum basale function as mechanoreceptors for light touch [4]. The layer directly below the epidermis, the dermis, is comprised of fibroblasts, vasculature, nerve fibers, sensory receptors, and extracellular matrix (ECM). The ECM is composed of two major components: collagen and elastic fibers. Collagen is the major structural component that provides both structure and tensile strength. Elastic fibers maintain tissue elasticity and resilience to restrain stretching [5] and are composed of elastin and microfibrils. The deepest layer, the hypodermis, is located under the dermis and composed of loose connective tissue with larger blood vessels, subcutaneous fat, and areolar connective tissues that provide insulation, thermoregulation, and cushioning to the skin [6].
As the body’s outermost layer, the skin is continuously subjected to external stressors such as solar radiation. Ultraviolet (UV) radiation from the sun can be categorized into three types based on wavelength: UVA (320–400 nm), UVB (280–320 nm), and UVC (100–280 nm) [7, 8, 9, 10]. The impact of UVC is minimal since the ozone layer completely absorbs it. However, the ozone layer only absorbs some UVA and UVB rays. This incomplete absorption allows a portion of UVB and UVA rays to reach the skin and penetrate the epidermis. Prolonged exposure to UVB can cause erythema and DNA damage in both keratinocytes and melanocytes, as well as stimulate proteolytic enzyme production. UVA is 10–100 times more abundant near the Earth’s surface than UVB and penetrates deeper, affecting both epidermal and dermal components (Fig. 2) [10, 11].
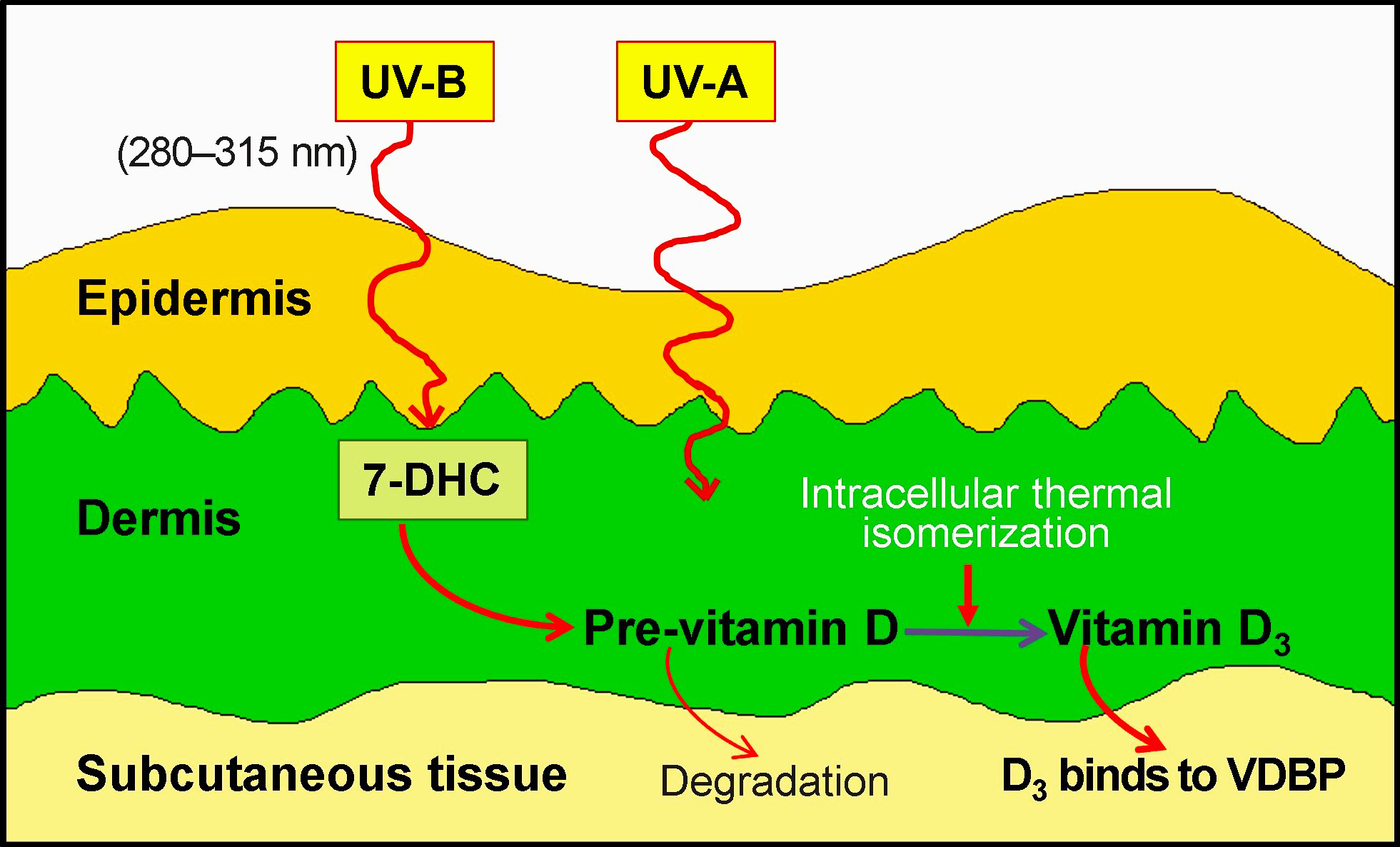 Fig. 2.
Fig. 2.
Wave-length-dependent ultraviolet (UV) radiation penetrates human skin and activates the production of vitamin D3 from the precursor 7-dehydrocholesterol (7-DHC).
UVB radiation converts 7-dehydrocholesterol (7-DHC) into pre-vitamin D, which gets photo-converted to vitamin D3 in the subcutaneous tissue (see Fig. 2). The cutaneous generation of vitamin D varies based on several factors, such as the amount of skin area exposed, intensity of sunlight, zenith angle of the sun, duration of exposure, skin type & thickness, and the application of sun-blocking chemicals [12]. Due to enhanced UVB absorption by melanin, individuals with dark skin have a lower capacity to generate vitamin D than those with lighter skin [13].
Vitamin D (calciferol) is a secosteroid molecule produced in the skin upon UVB exposure (Fig. 2). Following sufficient UVB exposure, 7-DHC is converted to pre-vitamin D3 in the epidermal basal and spinous cell layers. This pre-vitamin D3 gets converted to vitamin D3 (cholecalciferol) by thermal isomerization. The photosynthesized vitamin D3 and vitamin D2 (ergocalciferol) absorbed from the diet are fat-soluble compounds. They bind to the vitamin D binding protein (DBP) and are transported to the liver and other tissues via circulation [14]. In the hepatocytes, vitamin D2 and D3 are hydroxylated by the 25-hydroxylase enzyme (transcribed from CYP2R1), generating 25-hydroxycalciferol [25(OH)D], also known as calcifediol or calcidiol [15, 16]. Calcidiol is the predominant vitamin D metabolite in the bloodstream and the most accurate indicator of vitamin D nutritional status.
The DBP-bound vitamin D and 25(OH)D re-enter the circulation and are transported
to proximal renal tubular cells in the kidney and peripheral target cells [16].
In the kidney, 25(OH)D undergoes additional hydroxylation by the enzyme
1
The skin is the only organ capable of producing vitamin D, converting it to biologically active 1,25(OH)2D, and responding to it in a cell-specific manner [23, 26]. The metabolite 1,25(OH)2D is the high-affinity ligand of the transcription factor calcitriol receptor (used to known as vitamin D receptor, VDR). VDR regulates the expression of hundreds of target genes by binding to the vitamin D response elements on the chromosome [13, 14, 27, 28]. Under the direct influence of vitamin D, these target genes perform various skeletal and extraskeletal functions. A review by Carlberg C. [29] comprehensively covers the general overview of vitamin D signaling and VDR target genes.
In the skin, keratinocytes are the cells that respond to 1,25(OH)2D. VDR is most active in keratinocytes during cellular differentiation and proliferation [23], with the highest levels of VDR and CYP27B1 expression found in the stratum basale (basal layer) of the skin [30, 31]. Epidermal stem cells within the stratum basale produce transient amplifying cells, which begin the differentiation process as they ascend into the stratum spinosum. Once in the stratum spinosum, 1,25(OH)2D promotes the production of involucrin, keratin 1, and transglutaminase, which helps maintain proper barrier function [23].
The cells in the stratum granulosum contain profilaggrin, which is cleaved proteolytically to release filaggrin. In response to 1,25(OH)2D, cells in the stratum granulosum produce filaggrin and loricrin proteins, which together form the cornified envelope [23, 32]. Proteins play a crucial role in maintaining the integrity and function of the skin barrier [33].
The activation of genes by VDR and 1,25(OH)2D in the skin depends on the
level of cellular differentiation. Coregulators such as the Mediator complex
(MED, previously known as DRIP) and Steroid Receptor Coactivator (SRC) complex
(SRC2 and SRC3) modulate the transcriptional activity of VDR in a cell-specific
manner [34]. In undifferentiated cells, MED directly interacts with VDR to
inhibit its activity. In differentiated cell layers within the stratum
granulosum, the coactivators SRC2 and SRC3 enhance VDR activity [35]. Calcium
also influences the effects of 1,25(OH)2D and VDR indirectly by acting
through the multifunctional protein
The skin undergoes genetic, molecular, cellular, and organ-specific changes throughout a human’s lifetime [3, 38]. Intrinsic aging refers to the natural chronological changes that affect the entire human body over time. Extrinsic aging is defined as additional body changes exacerbated by environmental factors such as UV exposure and smoking. Fine wrinkles, thinning of the epidermis, reduced subcutaneous fat, and laxity are characteristic features of intrinsic skin aging.
In contrast, extrinsic aging involves additional changes exacerbated by environmental factors such as UV exposure and smoking [3, 8, 39, 40, 41]. As individuals age, intrinsic and extrinsic factors contribute to permanent skin modifications, resulting in thinner, dry, unevenly pigmented, and wrinkled skin with a sagging appearance [42, 43]. Fig. 3 (Ref. [44]) illustrates the characteristic features of young and aged skin.
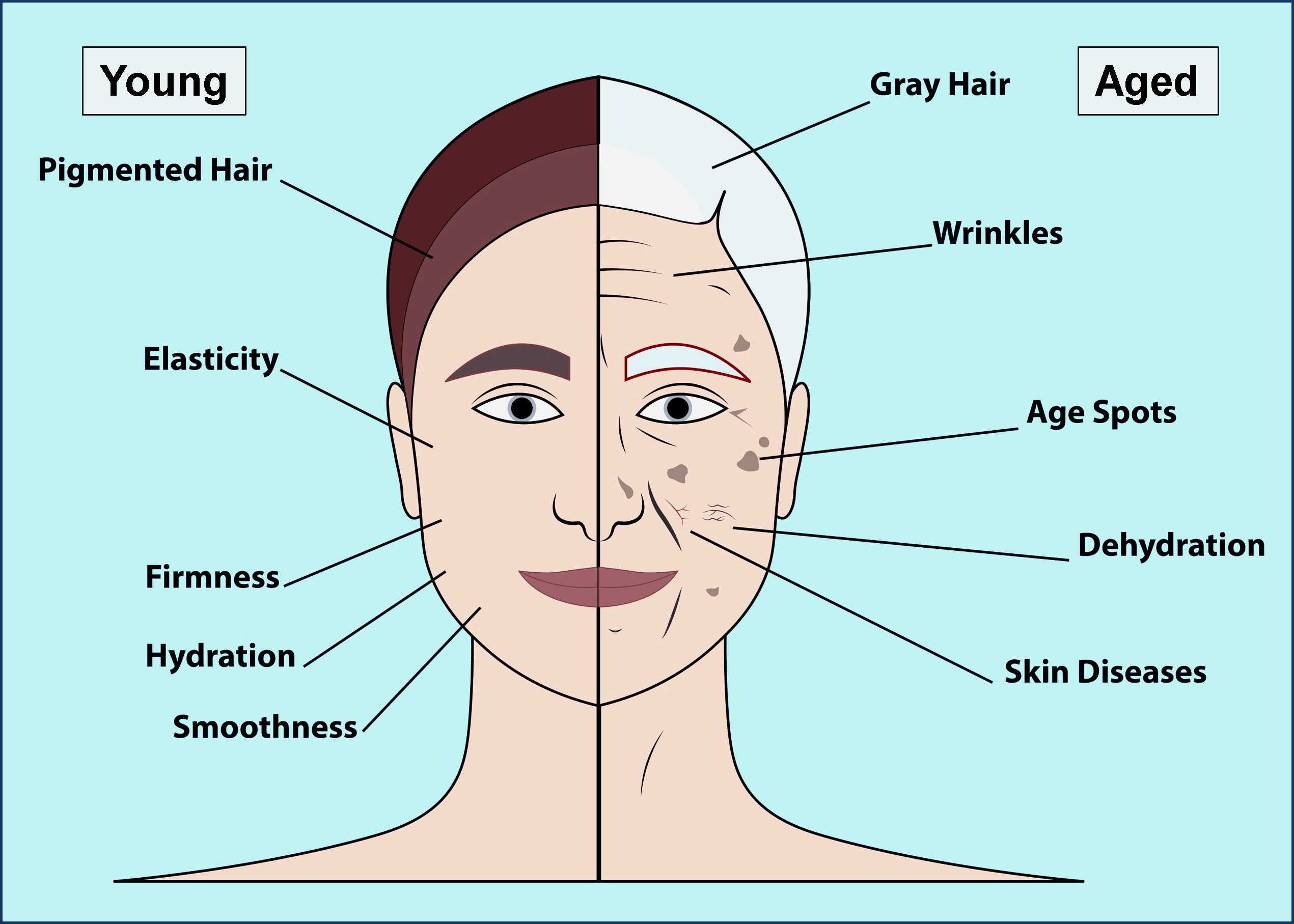 Fig. 3.
Fig. 3.
The characteristics of young and aged skin as visible to the naked eye (left side: young; right side: aged). Adapted from [44].
All layers of human skin undergo structural, physiological, and functional changes with age. The main structural changes in the epidermis include a reduction in thickness and changes in composition. The thinning of the epidermis is more noticeable in exposed regions of the body, with an overall reduction rate of 5–7% per decade. Decreases in dermal collagen and elastin, along with the reduction of subcutaneous fat in the hypodermis, are the major contributors to the overall thinning of the skin [45, 46]. With aging, the number of cells and epidermal turnover rate decrease, causing cells and tissues to undergo characteristic changes (Fig. 3).
Some age-related cellular changes in the epidermis include shorter and fatter keratinocytes, enlarged corneocytes, and reduced melanocytes, leading to uneven pigmentation. The number of Langerhans cells also declines with age, reducing antigen-trapping capability and hence impairing cutaneous immunity [47, 48]. Finally, subcellular changes due to aging also occur. For example, the dermo-epidermal junction, which provides strength, resistance, and nutrition to the skin, flattens with advanced age, increasing the skin’s vulnerability to damage from shear forces. The reduced nutrient and oxygen supply poses a risk for dermo-epidermal separation, contributing to wrinkles [45, 49].
Collagen, elastin, and hyaluronic acid, the main extracellular components of the dermis, all deplete in the aging skin (see Table 1, Ref. [40, 42, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68]). Dermal fibroblasts synthesize collagen, which strengthens and provides elasticity to the skin, and occurs in multiple forms. Type I collagen predominates in the skin’s connective tissue, followed by smaller amounts of type III [69]. Disorganized and fragmented collagen fibrils are a characteristic feature of the aged dermis. Collagen fiber bundles become stiffer and more rigid with age, creating rougher surfaces [50].
| Skin component | Observed change in skin | Reference |
| Epidermis | Reduced stratum corneum hydration | [51, 52, 53] |
| Dysfunction in epidermal permeability barrier | [54, 55] | |
| Dermo-epidermal junction flattens | [56] | |
| Decrease of the number of melanocytes | [57, 58] | |
| Decrease of the number of Langerhans cells | [57] | |
| Decrease of Merkel cells | [59] | |
| Elevation of skin surface pH | [60, 61] | |
| Dermis | Reduction of dermal fibroblasts | [62] |
| Decrease of collagen synthesis | [62, 63] | |
| Surface of collagen bundles become rougher, and fiber bundles become stiffer and harder | [50] | |
| Decrease of blood vessels and nerve endings | [40] | |
| Reduction of the thickness (dermis thinning) | [42] | |
| Decrease of elastic fibers | [64] | |
| Reduction of functional sweat glands and reduced sweat rate | [65] | |
| Decrease of Meissner’s corpuscles | [59] | |
| Sebaceous glands (SG) hyperplasia, SG atrophy, decreased sebum secretion, and altered sebum composition | [66] | |
| Decreased melanin production in hair shaft and reduction of bulbar melanocyte numbers | [67] | |
| Hypodermis | Decrease of the overall volume | [68] |
Elastic fibers, a highly durable macromolecular component of the ECM, provide elasticity to the skin and other tissues, but become fragmented and thinned with age, leading to reduced tissue elasticity [70, 71]. Oxidative damage, enzymatic degradation, calcification, carbamylation, aspartic acid racemization, formation of advanced glycation end products, and mechanical fatigue intrinsically lead to functional impairment of elastic fibers [71, 72, 73, 74]. Meanwhile UV-induced extrinsic aging fragments functional elastin and collagen, deposits non-functional elastotic material, and causes the accumulation of damaged elastin. All these effects contribute to solar elastosis [72, 75]. Table 1 further illustrates the changes associated with the structure of aged skin.
Oxidative stress contributes to both intrinsic and extrinsic aging processes.
The skin is constantly challenged with intrinsically generated reactive oxygen
species (ROS) through normal biological processes. ROS are independent molecules
with at least one oxygen (radical) atom and one or more unpaired electrons. They
include molecules such as hydroxyl radical (
External insults such as UV radiation, volatile and non-volatile chemical
pollutants, and smoking cigarettes further increase ROS production beyond the
levels generated by intrinsic factors. The skin undergoes oxidative stress when
the level of reactive species surpasses the neutralizing capability of its
endogenous antioxidant defense systems. The excess ROS can induce DNA mutations,
damage proteins, lipids, and carbohydrates, regulate inflammatory cytokines, and
trigger cell apoptosis [76, 78, 79]. ROS also affects other biological responses
and signaling pathways related to aging. These pathways include mitogen-activated
protein kinase (MAPK), nuclear factor kappa B (NF-
Skin cells contain multiple antioxidant substances and enzymes, which have protective functions to negate oxidative stresses. Compared to the dermis, antioxidant enzymes such as superoxide dismutases, peroxidases, glutathione reductases, and catalases display elevated activity in the epidermis. Antioxidant substances like alpha-tocopherol (vitamin E), ubiquinol-10, glutathione, uric acid, and ascorbic acid are also significantly higher in the epidermis. Other enzymatic antioxidants such as thioredoxin reductase, methionine sulfoxide reductase, heme oxygenase-1, and peroxiredoxins have also been observed in the epidermis and dermis [83, 84, 85, 86]. Given the enhanced exposure of the superficial layers of the skin to external insults, elevated levels of antioxidants in the epidermis are expected [87].
Intrinsic and extrinsic factors that cause DNA damage accelerate skin aging [88]. During chronological aging, skin cells produce more ROS, which oxidatively damage bases and lead to single-strand breaks and basic sites (apurinic/apyrimidinic site) in DNA [89]. Prolonged exposure of skin cells to ROS may influence the shortening of telomeres, further promoting cellular senescence [90]. Age-related reduction in DNA repair in the skin may also lead to the accumulation of damaged DNA in cells [91].
UV radiation predominantly causes the formation of cyclobutane-pyrimidine dimers (CPD) and 6-4 pyrimidine-pyrimidone (6-4PP) photoproducts in DNA [92, 93]. Additionally, UV radiation causes less common photoproducts such as Dewar valence isomers [94], purine photoproducts [93], and single and double-strand breaks in the phosphate backbone of DNA [95]. These DNA damages may disturb essential cellular functions, causing cell cycle arrest, apoptosis, or uncontrolled growth, leading to malignancy [92].
The tumor suppressor protein p53 functions as a transcriptional regulator modulating the expression of numerous genes that control cell death, senescence, cell cycle arrest, aging, DNA repair, carcinogenesis, and oxidative stress [96, 97]. p53 gets activated upon UVB-induced DNA damage and promotes G1 arrest in the cell cycle, providing time to recover from the DNA damage. Under weak to moderate oxidative stress, p53 protects the cell by stimulating antioxidant genes [98]. Irreversibly damaged cells will be eliminated by p53 through the triggering of apoptosis [99]. With these functions, p53 is critical in preventing skin cancers and recovering from skin photo damage. However, long-term skin exposure to UV radiation can cause mutations in the p53 gene. Mutated p53 has been observed in 50% of skin cancers, emphasizing the impact of these mutations [100, 101].
Persistent low-level systemic inflammation is a main contributor to the aging process. ‘Inflamm-aging’ describes the age-related activation of a persistent pro-inflammatory state via immune system dysregulation, which results in immuno-senescence [102]. Furthermore, inflammation plays a pivotal role in the degradation of ECM components, including elastin, collagen, and dermal hyaluronic acid, thus promoting the formation of wrinkles. Degraded collagen acts as a potent chemoattractant for immune cells, leading to increased inflammation [103].
UVR-induced DNA and ECM damage triggers cellular stress and activates
inflammatory responses in the skin. Cellular senescence from photoaged skin has
been detected in keratinocytes, fibroblasts, melanocytes, and subcutaneous
preadipocytes. Cells undergoing senescence arrest their proliferation and secrete
both anti-and pro-inflammatory mediators [104]. These mediators include
interleukins (IL-1, IL-3, IL-6, IL-8, IL-33), platelet-derived growth factor
(PDGF), various colony-stimulating factors (GM-CSF, M-CSF, G-CSF), high-mobility
group box 1 (HMGB1) protein, and transforming growth factor
Fragmented dermal collagen is a characteristic feature of aged skin. The low level of collagen production from aged fibroblasts and the age-related increase in collagen-degrading matrix metalloproteinases (MMPs) are the key contributors to the changes in collagen [50, 69, 110, 111, 112].
The MMPs constitute a group of zinc-dependent endopeptidases secreted by multiple cells, including keratinocytes and fibroblasts, and are responsible for breaking down nearly all components of the ECM. Based on the substrate specificity, the MMPs are divided into collagenases, gelatinases, stromelysins, and membrane-type MMPs [113, 114]. The collagenases (MMPs 1, 8, 13, and 18) degrade interstitial collagen I, II, and III; Gelatinase A (MMP-2) degrades type I and IV gelatin, whereas gelatinase B (MMP-9) degrades type IV collagens [113]. The activity of tissue inhibitor metalloproteinase-1 (TIMP-1), which inhibits many members of the MMPs, declines with advancing age [115], contributing to collagen fragmentation.
Finally, fragmentation of dermal collagen can occur due to internal factors stimulated by external and internal stresses. For example, during UV-induced photo-aging, ROS production and activation of cell surface receptors trigger the activation of MAP-kinase (mitogen-activated protein kinase) p38, JNK (c-Jun amino-terminal kinase), and ERK (extracellular signal-regulated kinase), which in turn recruit transcription factors like c-Fos and c-Jun. This cascade leads to the expression of the transcription factor AP-1 (activator protein-1) and induces the expression of collagen-degrading MMPs 1, 3, and 9 in fibroblasts and keratinocytes [116]. The observation of two elastolytic MMPs, matrilysin (MMP-7) and human macrophage metalloelastase (MMP-12), around elastotic material suggests the possible involvement of MMPs in the remodeling of elastotic areas in photodamaged skin [117].
Age-associated changes have been detected in epidermal proteins such as keratin 10, involucrin, pre-albumin, Hsp 27, and Rho B, which are involved in human keratinocytes’ differentiation and proliferation. These proteins are abundant in the epidermis of young individuals, but their expression diminishes with age and often becomes undetectable in older individuals [41]. Other mechanisms, such as epigenetic modifications [118] and the formation of advanced glycation end products [119], also significantly contribute to the skin’s aging process.
Vitamin D has exhibited protective roles, preventing intrinsic and extrinsic skin aging. These include inducing collagen production, regulating oxidative stress and inflammation, reversing the damage caused by UV radiation, increasing epidermal barrier function, and contributing to wound healing [77, 120].
Recent research has investigated the protective role of vitamin D against chronological and photo-aging of the skin. A study conducted by Chaiprasongsuk et al. [121] reported the potential of 1,25(OH)2D, cytochrome P450 11A1 (CYP11A1)-derived D3-hydroxyderivatives [20(OH)D3, 1,20(OH)2D3, 20,23(OH)2D3, 1,20,23(OH)3D3], lumisterol, and its hydroxy-derivatives [20(OH)L3, 22(OH)L3, 20,22(OH)2L3, and 24(OH)L3] to protect human epidermal keratinocytes against UVB-induced damage. Supplementation of any of the previously mentioned compounds at a concentration of 100 nM significantly reduced oxidant formation in human epidermal keratinocytes treated with 50 mJ/cm2 of UVB radiation [121]. In the treated keratinocytes, p53 phosphorylation at Ser-15 and nuclear localization of p53 were increased. The treatments also increased the expression of Nrf2-regulated antioxidant genes [121].
VDR plays a critical role in protecting the skin from UV-induced DNA damage. The impaired ability of VDR-null mice to remove 6-4 PP and CPDs and their susceptibility to UV-induced epidermal tumors suggests an association between VDR and DNA repair [38, 122]. In keratinocytes, VDR promotes nucleotide excision repair (NER) by facilitating the timely dissociation of Xeroderma Pigmentosum Complementation Group C (XPC) from the site of damaged DNA. This allows the timely assembly of downstream NER proteins and the pre-incision complex, thereby promoting DNA repair [123]. When keratinocytes are exposed to UV, vitamin D derivatives employ a variety of mechanisms to enhance DNA repair. These mechanisms include reduced reactive oxygen species, increased DNA repair and p53 expression, and higher energy allocation [124].
Vitamin D regulates genome-wide p53 signaling, protecting the cells against
photo-aging and skin cancers [101]. When treated with 1,25(OH)2D,
UV-irradiated skin cells produce increased levels of nuclear p53 (p
Apart from directly controlling gene expression, vitamin D exerts its regulatory effect by controlling the regulators of p53. One such example is the murine double minute (Mdm2) gene, a negative regulator of p53. Evidence suggests that 1,25(OH)2D and VDR receptors regulate Mdm2 gene expression in a p53-dependent manner [126]. However, future research investigating the indirect control of vitamin D on p53 regulators is required to solidify the novel regulatory pathways controlled by vitamin D in humans.
The enzymes superoxide dismutase (SOD) and catalase are key players in the cutaneous enzymatic antioxidant defense system. Studies show that SODs protect type I and type IV collagen from oxidative damage by interacting through the matrix-binding domain or heparin [127, 128]. The results of a longitudinal study comparing extracellular (EC)-SOD‒overexpressed transgenic and wild-type mice indicate that the expression of cutaneous EC-SOD gradually declines with advancing age. EC-SOD promotes collagen production in aged mice by activating adenosine monophosphate-activated protein kinase and nuclear factor erythroid-2 related factor 2 (Nrf2)/heme oxygenase-1 pathways, thus reducing skin aging [128]. Untari et al. [129] observed decreased SOD activity in vitamin D deficiency, suggesting a correlation between vitamin D levels and SOD activity.
Further, supplementation of vitamin D has increased the expression of the antioxidant defense enzymes SOD and glutathione peroxidase (GPX) in various human organ systems [130, 131]. However, vitamin D’s role in regulating these enzymes’ expression in human keratinocytes remains largely unknown, leaving an avenue for future research.
1,25(OH)2D-treated keratinocytes displayed a significant reduction
(p
MMPs degrade collagen, thus challenging the integrity of the skin. Recent
research has provided evidence for the protective role of calcipotriol against
MMPs. The dose-dependent suppression of mRNA and proteins in the MMP-9 and MMP-13
families by calcipotriol has been shown in a human squamous cell carcinoma (SCC)
cell line [133]. Similar experiments using other cell lines have provided
evidence of vitamin D’s protective role against MMPs. Stimulation of human lung
fibroblast (HFL-1) cells with interleukin-1
Li et al. [135] investigated the possible impact of vitamin D on
articular cartilage degradation by MMPs using the rat vitamin D deficiency model
and rat articular chondrocyte cells. Rat articular cartilage under vitamin D
deficiency displayed elevated MMP-9 and MMP-13 expression and treating with
1,25(OH)2D reversed this effect. Supplementation with TNF-
Recent research has investigated the protective role of vitamin D against autoimmune diseases. A randomized controlled trial involving 25,871 patients with autoimmune diseases observed that vitamin D supplementation over five years could reduce the risk of autoimmune diseases by 22% [136].
Calcitriol helps counteract the inflammatory responses caused by UV damage. It
reduces the levels of suppressive inflammatory cytokines such as TNF, IL-1,
IFN-gamma, and IL-2
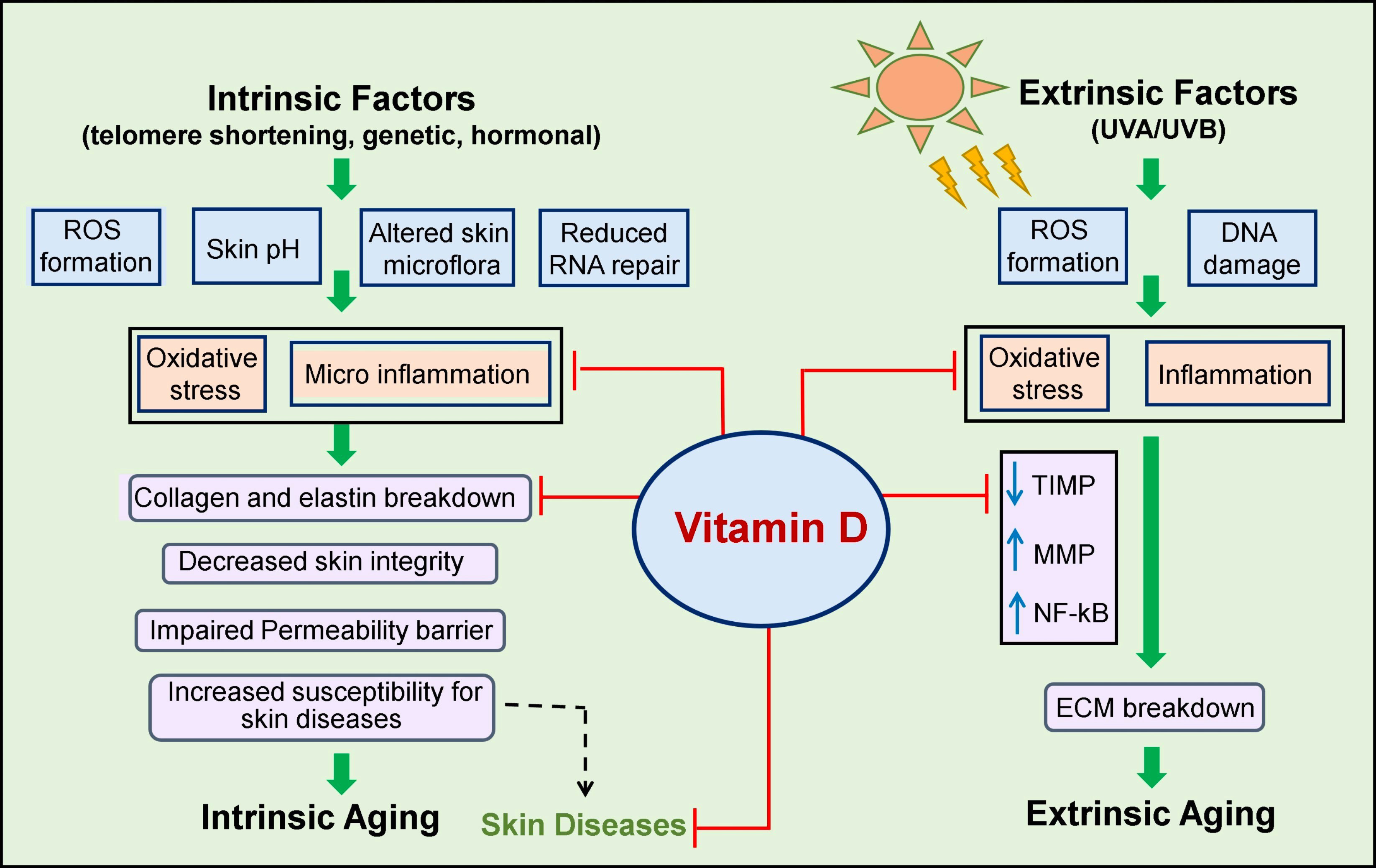 Fig. 4.
Fig. 4.
The impact of Vitamin D against intrinsic and extrinsic
skin aging and age-associated skin diseases. ECM, extracellular matrix; ROS,
reactive oxygen species; UVA, ultraviolet A; UVB, ultraviolet B; TIMP, tissue
inhibitor metalloproteinase; MMP, matrix metalloproteinase; NF-
With advancing age, the likelihood of developing skin conditions tends to increase. One study estimated that 50% of otherwise healthy older adults experience cutaneous diseases at some part of their adult life [141]. Cutaneous disorders can be painful, uncomfortable, and disfiguring, causing considerable physical and emotional distress in older people. They can negatively affect the appearance of already-aged skin, harming self-confidence and promoting self-isolation and social withdrawal. Skin diseases can also interfere with daily activities, decreasing the quality of life in aged populations [142]. The progressive immunosenescence contributes to the increased severity of infections associated with aging. The most common immunosenescence-associated diseases in geriatric populations are listed in Table 2 (Ref. [143, 144, 145, 146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156, 157, 158, 159, 160, 161, 162, 163, 164, 165]).
| Category | Condition/Disease | Source |
| Fungal infections of skin | Tinea pedis | [143, 144] |
| Candidiasis | [145] | |
| Fungal infections of nails | Onychomycosis | [146] |
| Onychauxis | [147] | |
| Inflammatory scaling dermatoses | Eczema | [148] |
| Contact dermatitis | [149] | |
| Seborrheic dermatitis | [150] | |
| Diseases of the dry skin | Xerosis | [151, 152] |
| Pruritus | [153] | |
| Vascular disorders | Pressure ulcers | [154] |
| Rosacea | [155] | |
| Cutaneous autoimmune disorders | Bullous pemphigoid | [156, 157] |
| Benign mucous membrane Pemphigoid | [158] | |
| Pigmentation disorders | Guttate hypomelanosis | [159] |
| Benign tumors | Seborrheic keratosis | [150] |
| Premalignant cutaneous lesions and skin cancers | Actinic keratosis | [160] |
| Basal cell carcinoma | [161] | |
| Squamous cell carcinoma | [162] | |
| Melanoma | [163, 164] | |
| Viral infections | Herpes zoster (shingles) | [165] |
The World Health Organization estimates that over 2 billion people worldwide suffer from deficiencies in essential vitamins and minerals [166]. Geriatric patients exhibit a higher prevalence of skin diseases and are generally low in serum vitamin D levels [167]. Recent studies have reported that vitamin D may play a role in protecting the skin against various diseases [168, 169, 170, 171].
Infections are common in aged skin; vitamin D regulates innate and adaptive
immunity, protecting the skin from infections. Calcitriol is essential for
adequately functioning immuno-modulating cells, such as T and B lymphocytes,
monocytes, macrophages, and dendritic cells [172]. It directly stimulates the
production of antimicrobial peptides, such as cathelicidin and
A continuous layer of microbes inhabits human skin. These microbes reside in the epidermis, dermis, and skin-associated glands and follicles, forming a diverse community known as the ‘normal skin microbiota’. Natural skin microbiome consists predominantly of bacterial species belonging to Actinobacteria, Firmicutes, Proteobacteria, and Bacteroidetes, and fungi belonging to Ascomycota and Basidiomycota [174]. Comparative studies have suggested age-related changes in the diversity and abundance of the skin microbiome and shown that any alterations to the native microbiota influence disease risk [175].
Cutaneous fungal infections are prominent among the aged populations. Factors including keratinocyte retention due to slower cell turnover, poor barrier function, and immunosenescence increase these infections [176]. Dermatophytosis, cutaneous candidiasis, and onychomycosis of the toenails are common fungal diseases affecting older people. Although direct studies investigating the role of vitamin D in combating fungal skin infections are lacking, there is a substantial amount of evidence proving the antifungal potential of vitamin D, as discussed below.
Vitamin D exhibits direct antifungal activity against the invasive fungus Cryptococcus neoformans. This fungus causes acute and chronic infections, including cryptococcal meningitis in humans. The broth microdilution assay indicates that vitamin D3 inhibits the fungus at a minimum inhibitory concentration (MIC 90) of 0.4 mg/mL. Vitamin D-induced intracellular ROS accumulation, altered cell membrane permeability, and compromised cell wall integrity in C. neoformans are the proposed mechanisms of fungal inhibition. Further, vitamin D reduces fungal cell adhesion and hydrophobicity while inhibiting biofilm formation in different C. neoformans developmental stages. The in vivo studies involving G. mellonella larval infection model demonstrated significantly reduced fungal burden in the vitamin D-treated group with enhanced survival of G. mellonella larvae [177].
Vitamin D3 also exhibits antifungal activity against several Candida species, including Candida albicans (ATCC 10261), C. krusei (ATCC 6258), C. tropicalis (ATCC 750), C. glabrata (ATCC 90030), C. dubliniensis (CBS 8501), and C. parapsilosis (ATCC 4344) with MIC ranging from 1–128 µg/mL. Further, it showed a significant reduction in Candida biofilm formation of up to 88% at 60 µg/mL with an IC50 value of 7.5 µg/mL [178]. According to a study by Lei et al. [179], vitamin D3 exhibited anti-Candida activity against standard and clinically isolated Candida species, with 90% growth inhibition achieved at a concentration of 0.4 mg/mL. The initiation, development, and maturation processes of biofilms in Candida albicans were also significantly affected. Reverse transcription-quantitative PCR analysis of vitamin D3 exposed C. albicans ATCC MYA-2876 cells proved the involvement of vitamin D3 in carbon metabolism, biogenesis of ribosomes, and biosynthesis of enzymes. A recent in vivo study conducted in IAC mouse models demonstrated that the fungal burden in the liver and kidney was reduced from day 3 to 14 with daily intraperitoneal treatment of high dose vitamin D3 (24,000 IU) [179].
A randomized, placebo-controlled clinical trial involving 416 pediatric patients between 12 months and five years has also provided evidence for the reduction of Candida infections upon supplementation of 300 IU/Day vitamin D in a yogurt drink. The prevalence of Candidaemia and Candiduria was significantly reduced in the vitamin D-treated group compared to the placebo group [180]. Finally, several studies have reported the role of vitamin D in enhancing host pulmonary resistance to the fungal pathogen Aspergillus fumigatus since vitamin D regulates the autophagy and Treg cells involved in A. fumigatus infection [181, 182, 183, 184].
Dry skin (xerosis) and pruritus are common dermatological conditions affecting most elderly people. It is speculated that pruritus affects virtually all individuals by the time they reach eighty years of age [185]. Chronic urticaria is another common allergic skin condition that causes itchy wheals lasting six weeks or more [186]. Multiple studies have examined the importance of serum vitamin D levels and the effects of vitamin D supplementation on these diseases [187, 188, 189].
In a retrospective case series, 57 patients with low vitamin D levels [25(OH)D
A randomized case-control study involving 192 patients with chronic urticaria
(CU) observed that vitamin D deficiency [25(OH)D
Atopic dermatitis (AD) is an inflammatory, pruritic skin disease commonly
occurring among pediatric and elderly (
Recent research has demonstrated a link between low serum vitamin D levels and atopic dermatitis. In a meta-analysis of eleven studies, a mean difference of –14 nmol/L 25(OH)D (95% CI –25 to –2) was observed in the AD patient group compared to healthy controls [192]. Another study showed that 84.3% of Bangladeshi children and young adults living in London with eczema had deficient or insufficient 25(OH)D levels. The research revealed a correlation between 25(OH)D deficiency and worse eczema severity among the study population [193].
Multiple studies have identified vitamin D supplementation as a promising
treatment for AD. A meta-analysis of three trials identified that the
supplementation of vitamin D significantly lowered scores on the scoring atopic
dermatitis (SCORAD) by 11 points (95% CI –13 to –9, p
Seborrheic keratosis is a common slow-growing, noncancerous skin growth, usually appearing in sun-exposed areas of the elderly population [169, 203]. SK is typically removed via surgery and other methods, including liquid nitrogen freezing and laser therapy. Various gene mutation studies and the utilization of topical vitamin D analogs such as Calcitriol evidence an association between vitamin D deficiency and SK [203, 204].
In a clinical study carried out by Mitsuhashi et al. [170], when 116 SK patients were treated with topical vitamin D3 ointments (tacalcitol 2 µg/g, calcipotriol 50 µg/g or maxa-calcitol 25 µg/g) for 3- to 12-months, 35 (30.2%) patients showed 100% or more than 80% decrease in the tumor volume, while 54 (46.6%) patients achieved a volume reduction between 40–80%. A Serbian study treating 12 patients with facial solitary seborrheic keratosis using 0.005% calcipotriol ointment for 3–8 months resulted in a complete regression of the lesions [169].
Finally, 30 adult SK patients (mean 52.7
Rosacea is an age-related chronic inflammatory skin condition mainly affecting
facial skin, characterized by transient or persistent redness (erythema),
telangiectasia, swelling, and lesions, including papules and pustules [207, 208].
Several studies reported an association between low vitamin D levels and the
occurrence of rosacea. A study conducted by Park et al. [209] observed
significantly reduced average serum 25(OH)D levels in rosacea patients than
healthy controls (12.18
Vitiligo is an autoimmune condition characterized by skin-depigmented white
patches due to epidermal melanocyte damage [213, 214]. Although the onset of
vitiligo is most common in childhood or young adults, late-onset has also been
reported in adults [215]. Cytokines play a crucial role in the development and
progression of this disease. Reduced vitamin D levels may significantly
contribute to the development of vitiligo by influencing Th1- and Th17-related
immune responses [216]. Many studies with controversial results have investigated
the association between vitamin D deficiency and vitiligo [217, 218, 219, 220, 221]. In a population-based
study, no significant difference was observed when serum vitamin D levels were
compared in patients with vitiligo and healthy controls [217]. Similarly, a
significant difference was not detected between the patients and controls in
studies conducted in Jordan [218] and Turkey [219]. Contrary to that, a
case-control study identified significantly lower vitamin D levels in 100 Indian
vitiligo patients (16.170
The efficacy of vitamin D supplementation as an option for enhancing the success
of laser-based treatments has been investigated in treating vitiligo.
Intramuscular cholecalciferol injection could enhance the therapeutic effects of
the excimer laser in vitiligo patients with low vitamin D levels (
Recent studies using in vitro cell lines, tissues, and animal systems have proven the protective role of vitamin D against various malignancies [225, 226, 227]. A daily intake of 1500 international units of vitamin D has been found to lower cancer mortality rates among men in the US by 30% [228]. However, conflicting results exist on the role of vitamin D against skin cancer. Patients who have acquired higher vitamin D levels through extended sun exposure are at an inevitably greater risk of developing skin cancer. This should be considered when evaluating the association between vitamin D and skin cancers.
The term ‘melanoma’ refers to aggressive malignant tumors originating from melanocytes. Upon exposure to UV radiation, genetic alterations activate oncogenes, deactivate tumor suppressor genes, and obstruct DNA repair. Consequently, unchecked growth of melanocytes occurs, causing melanoma [229].
The association between melanoma skin cancer and serum vitamin D levels is controversial. Many studies have identified an association between lower plasma vitamin D levels and (1) the development of melanoma, (2) an increase in the severity of the malignancy, and (3) a reduced survival rate in metastatic melanoma patients [230, 231, 232, 233, 234, 235].
In a retrospective study, higher 25(OH)D levels were associated with lower Breslow thickness of the melanoma at the diagnosis (p = 0.002) and better survival [236]. The highest survival was observed in the intermediate BMI group (24.9 to 29.9), which had the highest vitamin D levels (55 nmol/L). Previous research has observed the loss of vitamin D receptor expression in the cytoplasm and the nucleus of cutaneous melanoma (CM) cells. Vitamin D receptor expression negatively correlates with tumor progression; metastatic melanomas show the lowest cytoplasmic receptor staining out of all types of melanoma. The lack of VDR expression correlated to shorter survival of the patients [237, 238]. However, conflicting evidence also exists [239, 240]. A recent meta-analysis reported that each 12 ng/mL increment in 25(OH)D level was linked to a 42%, 30%, and 41% increase in cutaneous melanoma, keratinocyte cancer, and basal cell carcinoma, respectively [240]. Further evaluation is necessary to confirm whether increased safe sun exposure would reduce these risks.
Among skin cancers, non-melanoma skin cancers are the most common type of skin malignancy, particularly in fair-skinned populations of European descent. Most NMSCs are keratinocyte skin cancers, including basal cell carcinoma and squamous cell carcinoma [241]. Previous investigations into the role of vitamin D on NMSC have provided mixed results.
Basal cell carcinoma is the most common type of skin malignancy worldwide [242].
UVB radiation has been identified as the most critical risk factor for BCC,
especially for Fitzpatrick I, II, and other light skin types [243]. Based on a
study conducted in South Africa of BCC and SCC patients, 49.5% had deficient
(
In a comprehensive three-staged study involving 496 patients (mean age: 69
years), Ince et al. [171] observed that a serum vitamin D3 level
above 25 ng/mL could significantly reduce the recurrence rates of BCC. In a study
conducted with a cohort of Latvian patients with different primary and recurrent
BCC of the head and neck, 94.9% of patients were deficient in vitamin D.
Furthermore, an inverse association was observed between serum vitamin D levels
and tumor size [245]. When testing in the murine ASZ001 BCC line, potent
antitumorigenic activity was detected when 1,25(OH)2D,
1,20(OH)2D3, 1,20,23(OH)3D3, 1,20,24(OH)3D3,
1,20,25(OH)3D3 and 1,20,26(OH)3D3 hydroxy derivatives of
vitamin D were supplemented. The classical and CYP11A1-derived D3 hydroxy
derivatives, including 1,25(OH)2D3, 1,20(OH)2D3, and
20(OH)D3, inhibited the expression of glioma-associated oncogene 1 (GLI1)
and
However, controversial studies report increased BCC risk at high serum vitamin
D3 levels. Liang et al. [247] reported that the women with
In a systematic review and meta-analysis of BCC and cutaneous squamous-cell carcinomas (cSCC), Caini et al. [241] suggested a lack of a strong association between the serum vitamin D level and the NMSC risk. Moreover, a statistically significant association between 25(OH)D concentrations and cSCC was not detected among 1192 kidney transplant patients. Another study by de Gruijl et al. [250] observed a clear trend of higher 25(OH)D concentrations with the development of cSCC, possibly linked with the higher level of UV exposure promoting carcinogenesis. However, the intermittent supplementation of cholecalciferol has been proven helpful in enhancing photodynamic therapy-based treatments for SCC [251]. Therefore, further research is needed to uncover the potential of vitamin D supplementation as a monotherapy and/or in combination with other treatments for SCC.
The world population is aging, and there is a growing interest in understanding the physiological changes associated with advanced age. The skin undergoes intrinsic age-associated natural changes, such as telomere shortening, reduced DNA repair, oxidative damage to biomolecules, hormonal changes, immunosenescence, inflammation, and damage to ECM components. Extrinsic or environmental factors, such as UV radiation exposure can also cause DNA damage, oxidative stress, inflammation, ECM degradation, pigmentation changes, hyperkeratosis, and altered cell signaling, leading to “photo-aging”. Both intrinsic and extrinsic aging contribute together to the aged appearance of the skin. The physiological, biochemical, and functional changes that occur with aging also contribute to the high prevalence of skin diseases in older people. Both skin aging and cutaneous diseases can impose a physical and psychological burden on geriatric populations, reducing their quality of life.
Despite causing photo-aging, UVB radiation also induces the cutaneous synthesis of vitamin D. Numerous beneficial effects of vitamin D on human health have been reported over the past 15 years [12, 13]. However, the exact impact of this vitamin on skin aging remains poorly understood. Previous research has evidenced the potential of vitamin D derivatives to protect the skin by acting against various aging mechanisms, including oxidative stress, inflammation, activation of MMPs, and degradation of collagen and elastin. Furthermore, Vitamin D deficiency is a risk factor for various dermatological conditions. Studies suggest maintaining adequate levels of vitamin D to protect against the most common age-associated skin diseases. Maintaining the recommended vitamin D concentrations (above 40 ng/mL) via safe sun exposure or supplementation [17, 20] should be considered an effective and affordable strategy for maintaining overall skin health.
ECM, extracellular matrix; UV, ultraviolet; 25(OH)D, 25-hydroxyvitamin D; 1,25(OH)2D, 1,25-dihydroxyvitamin D/calcitriol; VDR, vitamin D receptor; CTR, calcitriol receptor; DBP, vitamin D binding protein; MPs, matrix metalloproteinases; ROS, reactive oxygen species; NF-
SSA: Conceptualization, writing-original draft, review and editing. GAA: Writing-original draft, conceptualization, review, and editing. SJW: Conceptualization, writing-original draft, review, and editing. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects.
Not applicable.
The authors would like to thank Dr. Jacob Hogins for reviewing and editing the grammar and language of the manuscript.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.




