The purpose of the study was to analyze the frequency of the spontaneous
posterior vitreous detachment (PVD) in patients admitted to an Emergency Eye
Department in Italy (EED) during the COVID-19 pandemic national lockdown in 2020
compared with the similar time period in 2019. In this retrospective
observational study, patient records for ophthalmology EED patients in the month
of April 2020 during the COVID-19 Italian national lockdown, were compared with
those for an equivalent one-month period in 2019. Diagnoses, gender, and age were
assessed. Unpaired Student t-tests were used for continuous variables.
Poisson regression was used for count analysis to compare categorical variables.
Chi-square test was applied to asses proportion differences. In comparison with
the 2019 equivalent period, there was a significant decrease in the overall
number of EED visits and in the number of patients presenting with a spontaneous
PVD during the 2020 lockdown (–41.6% and –49%, respectively). During the 2020
lockdown, all diagnostic categories showed less patient admittance, however, the
proportions remained stable when considering the entire cohort. The proportion of
urgent visits was 90% in 2020 and 86% in 2019 (p = 0.66). The
proportion of EED patients affected by spontaneous PVD was comparable between the
two study periods (8.4% in 2020 vs. 9.6% in 2019, p = 0.34). Patients
presenting with spontaneous PVD in both periods were significantly older when
compared to patients with other pathologies (mean age of 63years in 2020 and
64years in 2019, p
The vitreous humor (or vitreous body) is the gelatinous substance that fills the space in the eye between the lens and the retina, making up more than 75% and thus the largest anatomic structure of the eye [1]. It is transparent and colorless, made up of 98% water and the remaining 1–2% of hyaluronic acid, salts, sugars and collagen-type proteins [2]. It is thanks to the composition of the vitreous humor that the eyeball retains its spherical shape.
Isolated or confluent opacities may appear which can cause reduced vision. If these opacities are distinct and mobile, they are called floaters and the symptom induced is known as myodesopsia. Floaters are characterized by the sensation of seeing through a transparent liquid in which objects of various shapes and types float that escape when trying to fixate them. They can appear in the form of dots, rings, spots, filaments, moving bodies, flying flies, cobwebs and other different shapes, which tend to be perceived when looking at a bright surface and in bright conditions, such as looking towards the sky, a white wall or sheet of paper or a computer screen with a light background, or at times can be visible even with closed eyes. Besides aging, the deterioration of the gelatinous microstructure of the vitreous body can also be more commonly seen in conditions such as myopia, ophthalmological surgery and laser treatment, eye trauma and inflammations, low fluid intake, metabolic and genetic disorders, menopause and certain types of medication [2].
Posterior vitreous detachment (PVD) can occur when the vitreous humor withdraws from its anatomical position, losing the normal and perfect adherence to the retina. The onset of PVD is often the result of the aging process that all human beings undergo and mostly asymptomatic, affecting more than 75% of the population over 65 years [3], thus not considered a real morbid condition and classified as spontaneous PVD. Vitreous degeneration related to aging tends to begin with liquefaction or synchysis of the gel component and the formation of collagen fibrils into irregular aggregates, forming pockets of lacunae. There is also a weakening of the vitreo-retinal adhesions causing a collapse of the vitreous body [4].
The posterior vitreous cortex undergoes an acute separation from the internal limiting membrane ILM of the retina during PVD, mostly because of liquefaction of the vitreous and the adhesion weakening between the ILM and vitreous posterior cortex [5, 6]. Uncomplicated PVD, which is the most common form, usually does not give rise sight threatening conditions. PVD usually due to aging, typically has a good prognosis. Floaters, symptoms and myodesopsia normally are less symptomatic within a few months. If, however, vitreo-retinal adherence remains during liquefaction, abnormal traction can lead to complications of the retina and vitreous, which include retinal detachment (RD), macular holes, hemorrhages, vitreo-macular traction syndrome, retinal tears, and macular pucker [4, 7]. Patients with acute signs and symptoms need to be examined. These symptoms include worsening of photopsia and/or floaters. These flashes are caused by the vitreo-retinal traction and pulling on the retina. The risk of damage upon the onset of acute symptoms can be as high as 47.65% [8, 9]. RD can arise after acute abnormal PVD due to the liquified vitreous that moves under the retina causing traction and a separation of the retinal layers [10, 11, 12]. Immediate surgery is needed in the presence of important RD to avoid irreversible vision loss and permanent damage of photoreceptors [13]. Acute PVD patients should undergo an ophthalmologic assessment that includes thorough retinal fundus examination.
Photopsia, which are perceived as flashes, occur when the vitreous body tugs on the retina. Every touch or pull of the retina generates an electrical signal that is sent to the brain, and is interpreted as a flash, a circle or a line of bright light. Floaters appear larger and more numerous after acute PVD. Symptoms related to an acute spontaneous PVD are the new onset or worsening of myodesopsia and/or photopsia. These patients are usually directed to the emergency eye departments (EED). In a small percentage of cases, vitreous detachment can cause damage to the retina, which include rupture of the retina and/or the retinal detachment (RD) or macular holes [14]. Several studies have reported that the incidence of retinal breaks after acute PVD is about 15–20%, while RD is about 5–10% of patients attending EED with PVD [14, 15]. Immediate examination of patients is thus of utmost importance in patients with acute onset symptoms of PVD to provide rapid laser and/or surgical treatment and follow-up examinations in patients with greater risks.
COVID-19 has caused millions of deaths worldwide due to coronavirus disease since the first severe infections reported in December 2019 in Wuhan, China [16, 17]. Global pandemic measures by the World Health Organization began in March, 2020 due to the high contagious nature, mortality and morbidity of the disease. A national lockdown was imposed by the Italian Government in 2020 during the months of March, April and May [18]. Restrictions included isolation at home unless otherwise needed and a halt of all elective activities in all hospitals. Ophthalmology departments exclusively provided ophthalmic examinations for emergency and chronic sight-threatening conditions. The lockdown restrictions, in addition to the pandemic fear and infection risk, all play an influence on patients seeking acute clinical care [19].
Studied in literature have reported significant reductions of patients presenting at EEDs in the lockdown period of 2020 when compared to the same period due to several factors including behavioral changes and fear of contracting the disease, financial and logistic factors implemented during the lockdown, decrease of work accidents related and transportation, etc. During this particularly new and worrisome setting, numerous patients may have tended to underestimate symptoms or seek alternative or private healthcare systems. Poyser reported a 53% EED attendance decrease during the United Kingdom lockdown period [20]. A similar study reported a reduction of greater than 50% of ophthalmology emergency and acute services in London during the lockdown period [21]. Studies by Babu showed a 96.5% decrease of outpatient visits in India during the 2020 lockdown [22]. A Canadian retrospective study showed the total number of referrals decreased by 54.2% in the COVID-19 period versus the pre-COVID-19 period [23].
PVD can cause retinal detachment and ruptures, which can lead to irreversible visual loss if not treated timely in an appropriate clinical manner, thus immediate clinical ophthalmic examination is of utmost importance. The purpose of our study was to briefly review the current literature regarding PVD and to analyze the impact of the COVID-19 pandemic lockdown by assessing patients admitted to an Italian EED with acute PVD and other types of ocular pathologies in the month of April 2020 when compared with a similar non-pandemic 2019 time period.
All patients admitted to the EED at the University Hospital of Udine, Italy between April 1 to 30, 2020 were considered retrospectively for this observational study. Similar data was collected for the same period in 2019.
The study was conducted in accordance to the requirements of the Institutional Review Boards (IRB), HIPAA and tenets of the Helsinki’s Declaration. Informed consent was obtained before emergency evaluation.
Clinical records of all patients attending the EED of the University Hospital of Udine between April 1to April 30, 2020, during the COVID-19 pandemic lockdown in Italy, and the equivalent 2019 period were evaluated retrospectively. Extracted electronic data was comprised of: diagnostic code using ICD-9-CM 9th Revision of the International Classification of Diseases, Clinical Modification, date of admittance, gender and age. Patients were divided according to 9 diagnostic categories, which are reported in the appendix. The assessment included a comparison between the two time periods based on diagnosis, gender, age, and need for emergency follow-up care. The 14 subgroups included in the “vitreo-retinal pathologies” category were considered separately and, in particular, the data relative to the cases of spontaneous PVD were focused.
The Kolmogorov-Smirnov test was used to analyze the data distribution.
Continuous variables comparisons were assessed with the unpaired Student
t-test. Count analysis was performed with Poisson regression. Fisher’s
exact test and chi-square test were used for differences between proportions
Assessments were performed with SPSS Software (version 21.0, IBM Corp., Chicago, IL, USA). p
The number of patients assessed at the EED during the two study periods, which
are shown in Fig. 1, were respectively 297 (10
 Fig. 1.
Fig. 1.The average number of patients presenting at the Italian EED during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019.
Fig. 2 shows the total number of patients affected by spontaneous PVD in 2019
and 2020. Patients affected by PVD were respectively 25 during April 2020, and 49
in 2019, showing a 49% significant decrease in the lockdown period (P
 Fig. 2.
Fig. 2.The average number of patients presenting at the Italian EED with posterior vitreous detachment (PVD) during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019.
Fig. 3 shows the distributions for age and gender for the 2019 and 2020 periods.
The mean age for each period were not statistically different, and resulted to be
54
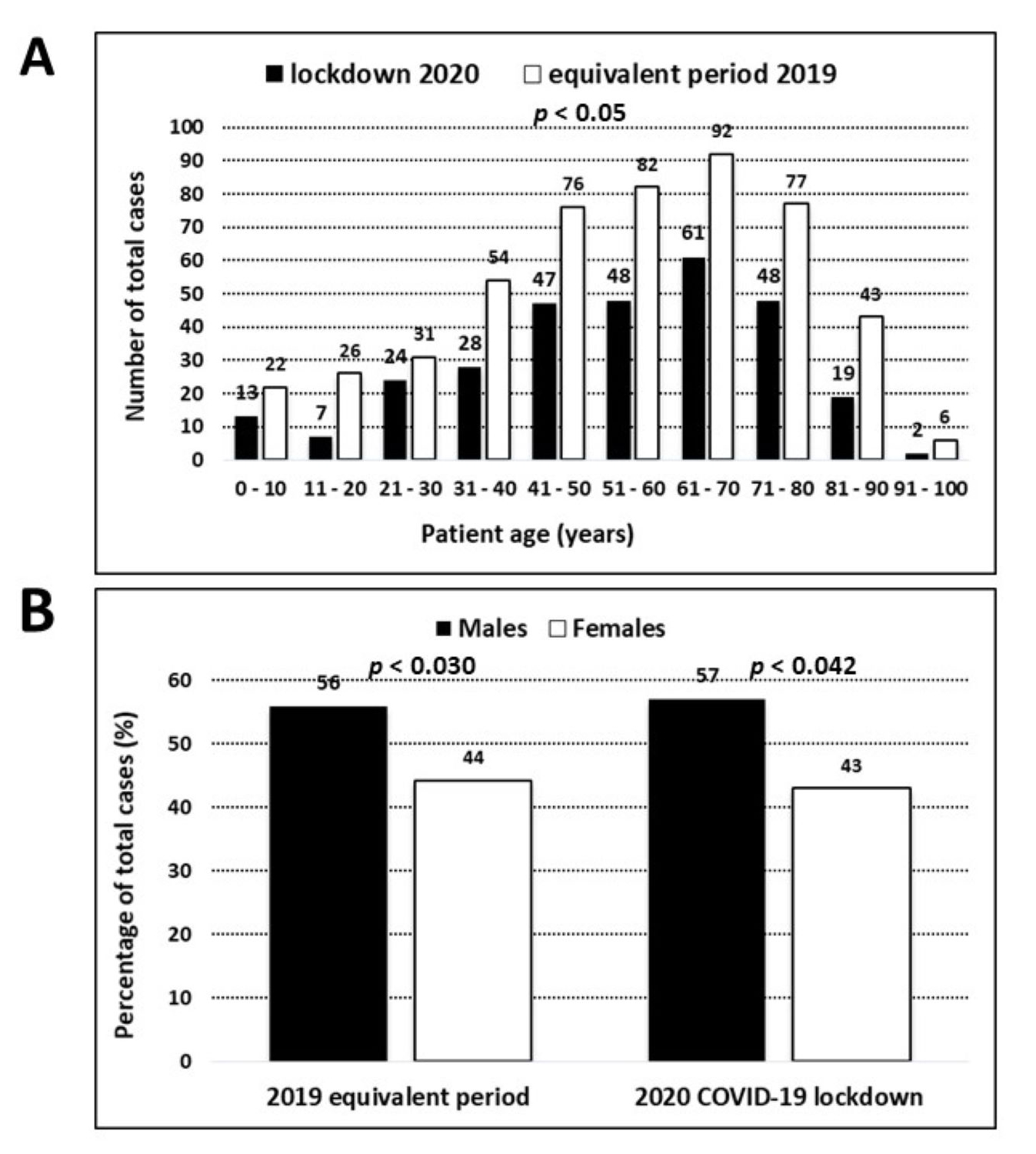 Fig. 3.
Fig. 3.The age and gender distribution of patients. (A) The age distribution of patients (based on age rank) presenting at the Italian EED during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019. (B) The gender distribution (% of total cases) of patients presenting at the Italian EED during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019.
There were significantly more males during both the 2020 lockdown (56.6% vs.
43.4%, p = 0.042) and during the same 2019 period (55.9 vs. 44.1%,
p = 0.03) (Fig. 3B). Males were slightly younger than females in 2020
(mean age of 53.1
Fig. 4 shows the distributions for gender and age for patients with PVD in 2019
and 2020. The mean age for patients was not statistically different (p =
0.54, unpaired Student’s t-test), and respectively were 63
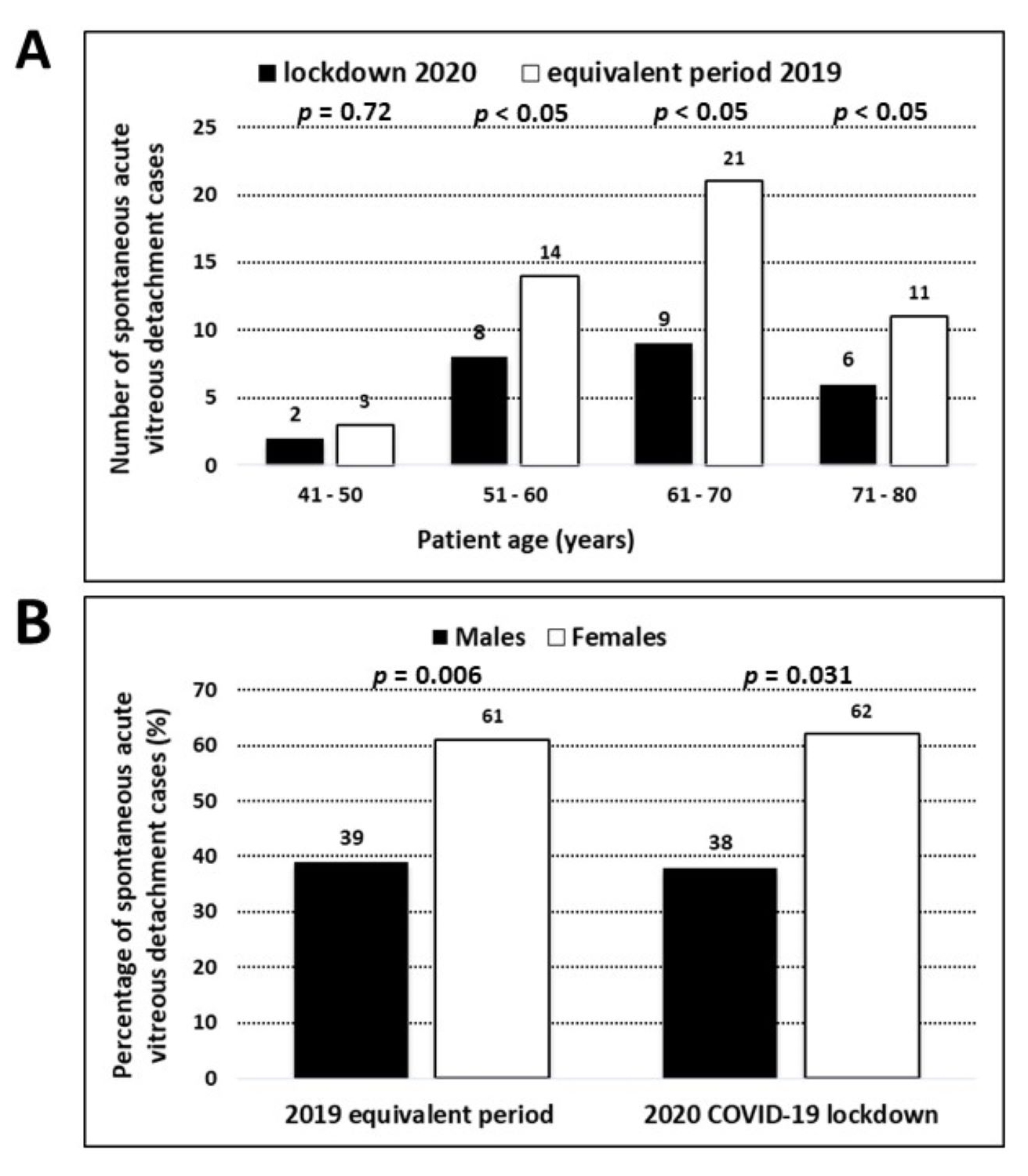 Fig. 4.
Fig. 4.The age and gender distribution of patients. (A) The age distribution of patients (based on age rank) presenting at the Italian EED with posterior vitreous detachment (PVD) during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019. (B) The gender distribution (% of total cases) of patients presenting at the Italian EED with posterior vitreous detachment (PVD) during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019.
Amongst patients having PVD, the percentage of females was significantly higher
than that of males both in 2020 (60% vs. 40%, p = 0.031) and in 2019
(61.2% vs. 38.8%, p = 0.006) (Fig. 4B). Males were slightly younger
than females in 2020 (mean age of 61.2
Table 1 shows the total number of patients for all pathologies divided in 9 diagnostic categories, in addition to gender distribution and mean age for 2020 and 2019. The pathologies most commonly seen in the EED during 2019 and 2020 were inflammations and traumas. In comparison with the equivalent period of 2019, during the 2020 lockdown the number of patients presenting at the EED decreased in all diagnostic categories except for the wet age-related macular degeneration (6 cases in 2019 vs. 11 cases in 2020) and for the rhegmatogenous retinal detachment subgroup (3 cases in 2019 vs. 7 cases in 2020). The spontaneous acute PVD, with 25 cases in 2020, and 49 cases in 2019, represented the most frequent vitreo-retinal pathology presenting at the EED during both 2020 and 2019.
| 2019 equivalent period | 2020 COVID-19 lockdown | Inter-group differences | |||||||
| (1–30 April 2019) | (1–30 April 2020) | 2020 minus 2019 | 2020 vs. 2019 | ||||||
| Diagnoses | cases | Males/Females | age (years) | cases | Males/Females | age (years) | n° of cases | n° of cases | |
| n° | (%) | (mean |
n° | (%) | (mean |
(% of total cases of 2019) | (p value)§ | ||
| Total number of cases | 509 | 55.9/44.1^ | 54.1 |
297 | 56.6/43.4^ | 54.0 |
–41.6 | ||
| Vitreo-retinal pathologies | |||||||||
| Posterior vitreal detachment | 49 | 38.8/61.2^ | 64.2 |
25 | 40/60^ | 63.0 |
–49.0 | 0.04 | |
| Vitreal floaters | 7 | 57.1/42.9 |
50.6 |
2 | 50/50 |
43.5 |
–71.4 | n.a. | |
| Retinal ruptures/degenerations | 9 | 66.7/33.3 |
59.4 |
3 | 66.7/33.3 |
55.7 |
–66.7 | n.a. | |
| rhegmatogenous retinal detachment | 3 | 100/0 |
59.7 |
7 | 57.1/42.9 |
58.3 |
+133.3 | n.a. | |
| Traction retinal detachment | 2 | 100/0 |
80.5 |
1 | 100/0 |
76 |
–50 | n.a. | |
| Macular pucker | 1 | 0/100 |
76 |
0 | / | / | –100 | n.a. | |
| Macular hole | 0 | / | / | 0 | / | / | n.a. | n.a. | |
| Dry age-related macular degeneration | 0 | / | / | 0 | / | / | n.a. | n.a. | |
| Wet age-related macular degeneration | 6 | 50/50 |
67.1 |
11 | 27.3/72.7^ | 73.6 |
+83.3 | n.a. | |
| Other causes of choroideal neovascularization | 1 | 0/100 |
67 |
1 | 100/0 |
54 |
0.0 | n.a. | |
| Diabetic retinopathy | 1 | 0/100 |
84 |
1 | 0/100 |
75 |
0.0 | n.a. | |
| Other causes of macular oedema | 0 | / | / | 0 | / | / | n.a. | n.a. | |
| Central serous chorioretinopathy | 1 | 0/100 |
41 |
0 | / | / | n.a. | n.a. | |
| Retinal dystrophies | 0 | / | / | 0 | / | / | n.a. | n.a. | |
| Inflammations | 161 | 46.5/53.5 | 56.0 |
98 | 47/53 | 47.8 |
–39.1 | 0.03 | |
| Traumas | 175 | 69.1/29.9^ | 47.6 |
97 | 72.4/27.6^ | 45.7 |
–44.6 | 0.02 | |
| Neurological disorders | 29 | 53.8/46.2 | 43.5 |
16 | 55/45^ | 46.0 |
–44.8 | 0.08 | |
| Degenerative pathologies | 21 | 53/47 | 66.1 |
14 | 50/50 | 68.5 |
–33.3 | 0.38 | |
| Open-angle glaucoma | 11 | 45.4/54.6^ | 74.1 |
4 | 25/75 |
72.5 |
–63.6 | n.a. | |
| Acute angle-closure glaucoma | 4 | 50/50 |
71.1 |
0 | / | / | –400 | n.a. | |
| Vascular pathologies | 7 | 65/35 |
67.6 |
5 | 60/40 |
67.6 |
–28.6 | n.a. | |
| Tumors | 6 | 50/50 |
57.2 |
3 | 66.6/33.3 |
58.1 |
–50.0 | n.a. | |
| Others | 14 | 58/42^ | 43.0 |
9 | 60/40^ | 46.1 |
–30.8 | 0.95 | |
| SD, standard deviation; ^, effect of gender on the diagnosis, statistically significant (Poisson regression for count); ^^, effect of age on the diagnosis, statistically significant (Poisson regression for count); §*, Poisson regression for count; *, statistically significant difference in the number of cases between study periods; n.a., not applicable for low number of cases. | |||||||||
In both study periods, older age and female gender appeared to be significantly
associated with PVD; conversely, male gender and younger age were risk factors
for trauma-related diagnoses. Furthermore, in both study periods, older age was
associated with glaucoma, degenerative and vitreo-retinal pathologies (Table 1),
whereas younger age was a risk factor for neurological disorders (p
Table 2 and Fig. 5 shows counts and histograms for all diagnostic for both time
periods. The percent of patients with trauma-related diagnoses and eye
inflammations were significantly higher than those of the other diagnostic
categories in both study periods (p
| Diagnoses | 2019 | 2020 | test* | |
| % of total cases | p value | |||
| Vitreo-retinal pathologies | ||||
| Posterior vitreal detachment | 9.6 | 8.4 | 0.34 | |
| Vitreal floaters | 1.4 | 0.7 | 0.50 | |
| Retinal ruptures/degenerations | 1.8 | 1.0 | 0.55 | |
| rhegmatogenous retinal detachment | 0.6 | 2.3 | 0.043^ | |
| Traction retinal detachment | 0.4 | 0.3 | 1.0 | |
| Macular pucker | 0.2 | 0.0 | 1.0 | |
| Macular hole | 0.0 | 0.0 | n.a. | |
| Dry age-related macular degeneration | 0.0 | 0.0 | n.a. | |
| Wet age-related macular degeneration | 1.2 | 3.7 | 0.021^ | |
| Other causes of choroideal neovascularization | 0.2 | 0.3 | 1.0 | |
| Diabetic retinopathy | 0.2 | 0.3 | 1.0 | |
| Other causes of macular oedema | 0.0 | 0.0 | n.a. | |
| Central serous chorioretinopathy | 0.2 | 0.0 | n.a. | |
| Retinal dystrophies | 0.0 | 0.0 | n.a. | |
| Inflammations | 31.6 | 33.0 | 0.26 | |
| Traumas | 34.4 | 32.7 | 0.31 | |
| Neurological disorders | 5.7 | 5.4 | 0.49 | |
| Degenerative pathologies | 4.1 | 4.7 | 0.74 | |
| Open-angle glaucoma | 2.1 | 1.3 | 0.10 | |
| Acute angle-closure glaucoma | 0.8 | 0.0 | 0.30 | |
| Vascular pathologies | 1.4 | 1.7 | 0.66 | |
| Tumors | 1.2 | 1.0 | 0.71 | |
| Others | 2.5 | 3.0 | 0.47 | |
| *, chi-square test o Fisher’s exact test; ^, statistically significant. | ||||
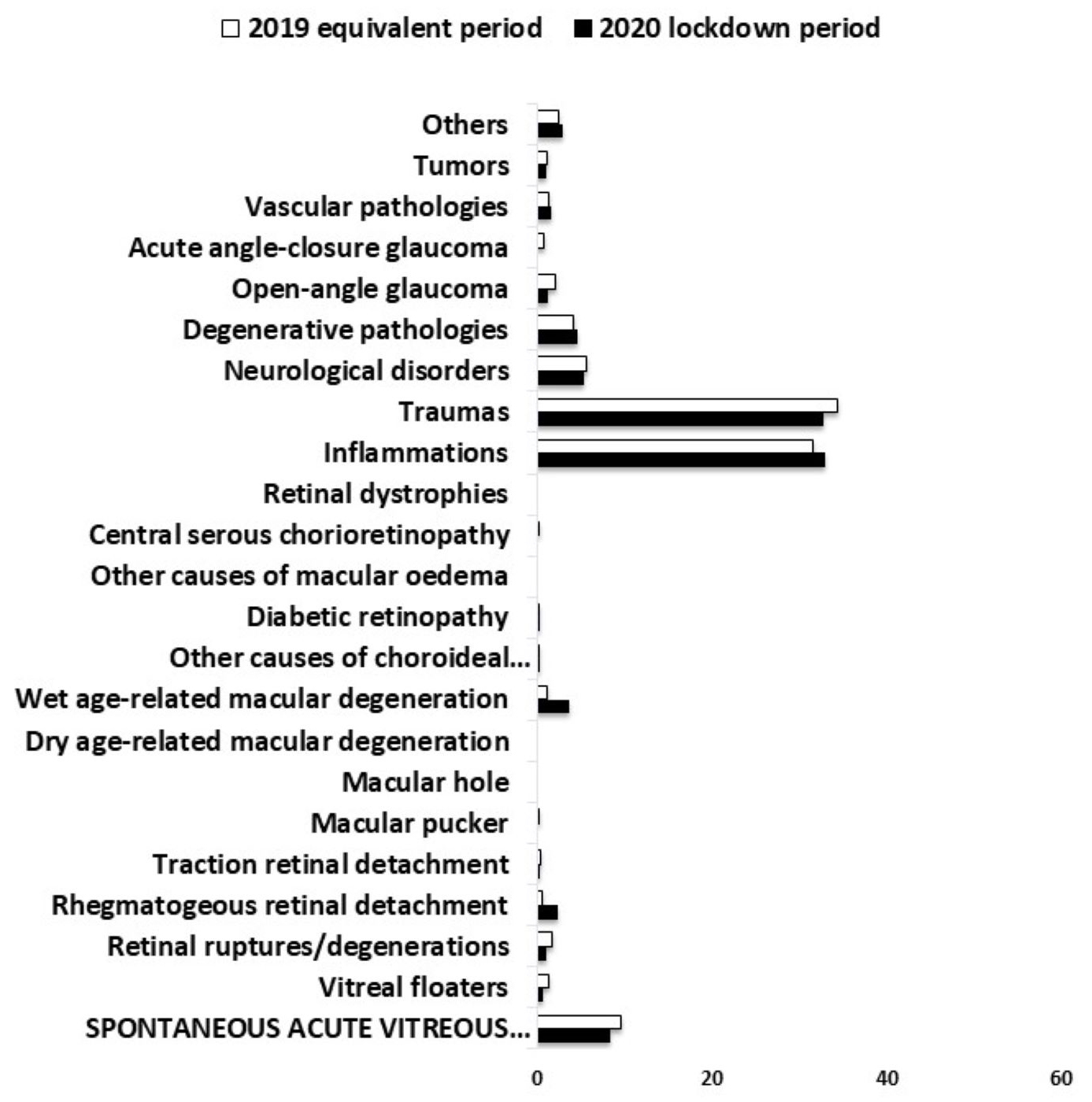 Fig. 5.
Fig. 5.The most frequent diagnoses (% of total cases) of patients presenting at the Italian EED during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019 (* = statistically significant difference between groups).
Patients defined as non-urgent after thorough EED examination (Fig. 6) were slightly less in 2020 (10%) when compared to 2019 (14%) (p = 0.34, chi-square test). After EED examination, further emergency measures were performed in 28 cases with acute visual loss in 2020 (Table 3), which increased slightly to 31 in 2019 (p = 0.84, Poisson regression for count). All patients attending the EED included in the study cohort and hospital staff showed no symptoms or signs of coronavirus disease.
 Fig. 6.
Fig. 6.The percentages of patients presenting at the Italian EED classified as urgent or non-urgent during the 2020 COVID-19 lockdown (April 1–30, 2020) in comparison with the same period in April, 2019.
| Procedures | 2019 equivalent period | 2020 COVID-19 lockdown | p value* | |
| (April 1 to April 30, 2019) | (April 1 to April 30, 2020) | |||
| (number of cases) | (number of cases) | |||
| Laser barrage for peripheral retinal breaks | 9 | 3 | 0.06 | |
| Panretinal photocoagulazion for proliferative diabetic retinopathy | 1 | 1 | n.a. | |
| Laser iridotomy for acute angle closure attack or occludible angle | 3 | 0 | n.a. | |
| Intravitreal anti-VEGF injection for: | ||||
| neovascular glaucoma | 1 | 0 | n.a. | |
| active choroideal neovascular membrane | 7 | 12 | 0.08 | |
| ischemic retinal vein occlusion | 4 | 2 | n.a. | |
| diabetic macular oedema | 1 | 0 | n.a. | |
| Hospitalization for ocular surgery in: | ||||
| retinal detachment | 5 | 8 | 0.15 | |
| corneo-scleral sutures for traumatic eye perforation | 0 | 1 | n.a. | |
| amniotic membrane application for corneal ulcer | 0 | 1 | n.a. | |
| Total number of cases | 31 | 28 | 0.71 | |
| *, Poisson regression for count; VEGF, vascular endothelial growth factor; n.a., not applicable for low number of cases. | ||||
The pandemic period in 2020 due to COVID-19 imposed restrictions and a radical re-organization of public services and public hospitals to limit the transmission of the virus and enhance critical care to infected patients with severe symptoms. Factors that include social distancing measures, restrictive guidelines, limited access to hospital for emergency care, and patient fear of infection in public places during the lockdown period, all played a role in the important decrease of ophthalmic examinations and EED services. The balance between infection control during the pandemic period and proper ophthalmic patient management is important, considering that certain acute eye conditions can lead to irreversible visual function loss if there is a delay in treatment. PVD is one of these conditions that can cause irreversible vision loss due to acute RD, thus timely diagnosis and appropriate treatment with laser and/or surgery and follow-ups in patients at risk are of utmost importance. We wanted to study the impact of the COVID-19 pandemic period in 2020 on the access to the EED for PVD based on a one-month assessment during the 2020 lockdown compared to 2019.
Our results showed that there was a statistically significant decrease of cases at our EED of 41.6% when compared to the same month of April in 2019 (Fig. 1). The differences appeared higher in the first 10 days. The reduced volume in patients in the EED may be multifactorial, including factors related to all the national restrictions and social distancing guidelines imposed by the lockdown; decrease in trauma or work-related injuries due to less time spent in travelling, at work or on holidays; reorganization of territorial and hospital clinics; and, fear and physiological issues related to risks of infection in public areas [24, 25]. Our results are in accordance with numerous studies in literature that have reported a drastic reduction in hospital and ophthalmologic EED attendance worldwide during the lockdown period [20, 21, 22, 23, 24, 25, 26, 27]. With regards to spontaneous acute PVD, which represents a natural aging process, the incidence and prevalence are expected to be relatively stable and related to the aging population [3]. The significant reductions of cases of spontaneous PVD during the lockdown (Fig. 2) suggest that the patients may have neglected or underestimate symptoms like flashes, floaters or metamorphopsia.
The age distribution of patient attending the EED did not differ significantly in 2019 and 2020, showing a mean patient age of 54 years (Fig. 3, Table 1). The total number of patients was significantly lower for each age rank during 2020, with exception to patients older than 90 years. The portion of males was significantly higher in both periods, showing a male predominance of 56.9% and 55.9% in 2020 and 2019, respectively. The higher male bias in is accordance with previous studies [22, 27], which could be due to females that remain home to tend to children and family members and greater work-related incidents in traditional male-dominated manual labor positions.
With regards to patients diagnosed with spontaneous PVD at the EED, the mean ages were similar and not significantly different, resulting to be 63 and 64 years in 2020 and 2019, respectively (Fig. 4, Table 1), which is similar to data reported in other studies [15]. The majority of patients with PVD in both 2019 and 2020 aged between 50 to 70 years (Fig. 4), which is in accordance with the natural history of PVD with aging, that tends to begin at the age of 40, then gradually progresses in the next 2 decades, then virtually completed after the age of 70 [28, 29].
Our results show that females were more prone to PVD independently of the lockdown period, with an incidence of 60% and 61.2% in 2020 and 2019, respectively (Table 1, Fig. 4B). Women tended to be older in both time periods. This in accordance with sex-related differences in the PVD reported in literature, with a greater prevalence in females [8, 28, 30, 31]. Several studies have attributed gender effects due to different levels of estrogen, higher intake of vitamin B6, advanced glycation and differences in biochemical composition of the vitreous and longer average lifespans in women [2, 8, 30, 31, 32, 33, 34].
The most frequent pathologies found in our EED during the month of April in both 2019 and 2020 (Table 1) were: (1) inflammations, with 161 and 98 cases, respectively (especially blepharo-conjunctivitis and conjunctivitis). (2) eye trauma showing 175 and 97 cases, respectively (mostly external eye foreign bodies, abrasions of the cornea, and ocular contusions). (3) vitreo-retinal pathologies. With regards to the more prevalent vitreo-retinal pathologies reported, the number of patients found in our EED in 2019 and 2020 for the subgroups PVD, vitreal floaters, retinal degeneration, rhegmatogenous retinal detachment, traction retinal detachment and wet age-related macular degeneration were respectively: 49 and 25; 7 and 2; 9 and 3; 3 and 7; 2 and 1; 6 and 11 (Table 1), with a reduction of cases in 2020 for all vitreo-retinal pathology subgroups, except for the rhegmatogenous retinal detachment and the wet age-related macular degeneration cases, that showed an increase both in the number of cases (not significant, Table 1), and in the percentage of patients (statistically significant, Table 2). PVD showed the highest number of cases amongst all vitreo-retinal pathology subgroups (49 and 25 cases in 2019 and 2020, accordingly). Our results seem to suggest that the COVID-19 2020 lockdown restrictions did not have a significant impact on the range and percent of cases based on total caseload of pathologies presenting at the EED (Fig. 5). As reported in Table 1, although the number of patients examined drastically decreased from 509 to 297 (due to fear of infection, lockdown constrictions, and probable self-medication for pathologies with minor symptoms), the percentage of diagnosis based on total caseload did not differ significantly for most categories (Table 2).
With regards to the incidence of pathologies (Table 1), trauma-related diagnoses factors included younger age and male gender (probably due to a higher incidence of work-related minor traumas in male-dominated manual labor positions). In is interesting to note in literature that young men, mostly within the age of 20–40 years, constitute 92% to 100% of the patients presenting EEDs for intraocular foreign bodies [35, 36]. The 44% decrease of traumas during the lockdown period is in accordance with other studies that reported a drop of more than 50% due to a reduction of various work-related and leisure activities during the pandemic [37, 38]. Female gender and older age, on the other hand, appeared to be significantly associated with spontaneous acute PVD for both periods, in accordance to the greater prevalence of PVD in older females reported in literature [8, 28, 30, 31].
Our data showed a decrease in improper EED access in 2020. Patients having non-urgent cases were 14% in 2019, which decreased to 10% in 2020 (Fig. 6), which is indicative of patients probably seeking EED assistance mostly for acute important symptoms (that can turn out to be urgent cases) during the lockdown, thus neglecting vague or mild symptoms due to constrictive measures and fear. The number of patients needing emergency interventions after EED assessment was 31 and 28 patients in 2019 and 2020, respectively, which was quite similar and not statistically significantly different (Table 3).
The most typical symptoms for PVD are myodesopsia, floaters and photopsia (or flashes). Abnormal PVD arises when vitreal adhesions to the retina remain strong in the presence of liquefaction of the vitreous humor [4], which can give rise to retinal breaks and RD that can lead to irreversible loss of vision. Our results show that 9 patients underwent barrage laser in 2019, which decreased to only 3 cases in 2020 (Table 3), which could be indicative of patients misinterpreting acute floaters or flashes as important symptoms in the midst of fear and constraint during the lockdown period. It is interesting to note that the number of cases with diagnosed retinal detachment upon EED assessment increased from 5 in 2019 to 8 cases during the 2020 lockdown. This could be due to a portion of patients that did not seek prior assistance with the acute onset of mild photopsia and myodesopsia for acute PVD. The pandemic restrictive measures and fear of attending the EED may have outweighed preventative measures for vague symptoms that normally would cause patients to seek the EED in non-lockdown situations.
Studies have shown that photopsia and myodesopsia tend to be important elements in the formation of retinal tears. Retinal tear risk factors include the acute presentation or increase of numerous floaters and retinal or vitreous hemorrhage [10, 39, 40]. Acute PVD with retinal tears can cause RD, because fluid from the vitreous can position underneath the retinal break, thus causing separation of the retinal pigment epithelium from the neuroretina, especially if there is traction of the retina [10, 11, 12, 13, 14]. RD can develop before 6 weeks if retinal breaks are not treated, which tends to be more likely to occur and faster in the presence of multiple and/or new tears and in the presence of retina and/or vitreous hemorrhages [10, 14]. These signs and symptoms need urgent evaluation, with utmost importance in cases with greater risks of RD, which include high myopia, pseudophakia/afachia, previous retinal tears, trauma or laser treatment, history of ophthalmic surgery, etc. [10, 11, 12, 13, 14].
Barrage laser can help prevent RD, however, time plays an important role. Early treatment can only be considered if patients seek emergency ophthalmic evaluation, which may have been outweighed by fear. In 2019, preventative barrage laser was performed in 9 patients, while 5 patients had RD. These numbers seemed to be inverted in 2020, in which the 8 RD cases outnumber the preventative 3 patients that underwent laser treatment, probably reflecting the effects of the lockdown and fear in the delay in seeking an urgent ophthalmic evaluation. One may hypothesize that these 8 patients with RD could have been less if treated with barrage laser before the onset of RD, and maybe, several of these patients may have avoided the EED as an effect of the COVID-19 lockdown.
Our data is in accordance with current literature regarding the great impact of the COVID-19 pandemic on ophthalmological care [20, 21, 22]. This restrictive period has brought on numerous limitations in timely sight-saving therapies, causing deleterious consequences in terms of preventative care and visual disabilities. There were significantly less patients in 2020 that used the eye emergency service, especially for pathologies that were not urgent. Potentially vision threatening pathologies like RD due to abnormal PVD can be prevented or treated if diagnosed and assessed in a rapid and thorough manner. Patients need to be educated and informed on important ocular symptoms and signs, and seek urgent evaluation, even in the midst of restrictions and lockdown periods.
In conclusion, it is important to note that floaters and flashes are typical signs of acute PVD. Uncomplicated PVD arises with liquefaction of the vitreous body in the presence of weakening vitreo-retinal adhesion. Abnormal PVD, usually due to vitreo-retinal tractions, can lead to complications that include retinal tears, hemorrhages and RD. Patients with isolated and peripheral tears can be treated with barrage laser to avoid RD. It is thus very important to inform patients about important symptoms, especially if at risk, in which time is of the essence and can prevent irreversible vision loss.
MZ was responsible for the conception and design of the study, assisted in the writing, outline, final approval of the version of the article to be published and completed the English and scientific editing (a native English-speaking MD, PhD); MLS assisted in writing the paper and for the conception and design of the study, performed the statistical analysis, results and figures and tables; CS assisted in the writing, editing and making critical revisions of the manuscript.
The study was in compliance with the tenets of the Helsinki’s Declaration and with the Institutional Review Boards (IRBs) and HIPAA requirements. Due to the retrospective observational nature of the study, the IRBs waived the need for formal approval and informed consent from participants.
We would like to thank Leonardo Macor and Simonetta Del Bianco from the GESPEC Department at the University Hospital in Udine for the assistance with the diagnosis grouping and data retrieval system developed for the Udine data collection.
This research received no external funding.
The authors declare no conflict of interest.
