1 Department of Urology, Salford Royal NHS Foundation Trust, M6 8HD Salford, UK
2 Department of Urology, Haukeland University Hospital, 5021 Bergen, Norway
3 Department of Urology, University Hospital Southampton NHS Foundation Trust, SO16 6YD Southampton, UK
Abstract
Kidney stone disease (KSD) is a common urological condition. For the patient, it typically presents with severe colicky flank pain and can have a substantial impact on quality of life, work productivity, and livelihood. For the clinician, they can present a diagnostic conundrum particularly in the absence of appropriate imaging, and the cost burden to the healthcare system can be significant. Investigations serve to rule out underlying metabolic, structural or pathological abnormalities, as well as identify the size, number, location, type and associated features of stone. KSD treatment can be complex, encompassing a spectrum of conservative, dietary, medical, and surgical approaches. This article provides an overview of the risk factors, clinical features, diagnosis, prevention and management of KSD. We conclude with a review of the recent innovations in this field.
Keywords
- stone
- urolithiasis
- kidney calculi
- laser
- ureteroscopy
- percutaneous nephrolithotomy
Urolithiasis affects millions globally and can lead to significant morbidity
including severe pain, urinary tract obstruction and sepsis. With an estimated
prevalence of between 1% and 13% across different regions of the world
[1], it has become a subject of interest not only due to
its rising prevalence but also the increase in the number of interventions, which
come at a significant cost to the healthcare system [2].
The epidemiology of kidney stone disease (KSD) varies significantly based on
geographic, socio-economic, and climatic factors. Obesity and metabolic syndrome
among others, are recognized as predisposing factors for KSD [3]. Based on population growth and trends in metabolic syndrome, the cost of
managing KSD in the USA in the year 2030 has been estimated at
KSD research is faced with various challenges including lack of understanding of the link between genetics and environmental factors, limited understanding of the mechanism behind stone nucleation and growth, developing effective preventive measures for recurrent stone formers with complex metabolic profiles, and addressing disparities in access to care based on socioeconomic and racial factors. In this paper, we discuss the pathogenesis, risk factors, treatment modalities and innovations in KSD management.
The first step in stone formation is supersaturation which occurs when the concentration of certain solutes in urine exceeds their solubility [6]. Excess solutes begin to aggregate, forming crystals that further combine to form kidney stones. The degree of supersaturation in addition to other factors such as crystallisation inhibitors and promoters, urinary pH and urine volume play a significant role in stone formation. Nucleation occurs when crystals form from a precursor (a nucleus) in the presence of supersaturated urine. One major stone nucleus is Randall’s plaque—a calcium phosphate deposit formed in the basement membrane of the thin limb of the loop of Henle. These plaques increase in size, rupture through the urothelium over the renal papilla and into the renal calyces [7]. Calcium oxalate layers begin to form on this calcium phosphate substrate, aggregating with other crystals to form kidney stones. Struvite stones are formed by urease-producing bacteria which split urea and promote ammonia and carbon dioxide formation leading to urine alkalinisation and formation of magnesium ammonium phosphate stones [7]. Anatomical abnormalities such as polycystic kidney disease, medullary sponge kidneys and urinary tract diverticula are risk factors for kidney stone formation, likely due to urinary stasis in addition to the already described lithogenic modifiers [8].
Several factors predispose patients to kidney stone formation. An understanding of these factors and their mechanisms is vital in identifying high risk individuals, planning targeted health education and promotion measures, as well as early prevention and treatment of affected patient groups, thus reducing the global burden of this disease. Risk factors for KSD and their mechanisms are outlined in Table 1 (Ref. [9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20]).
| Risk factors | Mechanism |
| Age | likely due to altered urine composition and increased comorbidities with age [9]. |
| Gender | higher stone prevalence in men could be attributed to differences in urinary tract anatomy and hormone profile [10]. |
| Diet | excessive consumption of foods rich in oxalate, urate, sodium, and animal protein increases the risk of stone formation [11]. |
| Chronic dehydration | low urine output occasioned by inadequate fluid intake can increase the concentration of urinary salts and lead to lithogenesis [12]. |
| Obesity | obesity increases the risk of urate and/or calcium stones due to its connection with acidic urine, uric acid stone and hypercalciuria [13]. |
| Environmental factors | hot and arid climates increase the risk of stone formation due to its impact on hydration and urine volume. Chronic lead and cadmium exposure have also been shown to be risk factors [14]. |
| Family history | a positive family history of kidney stones is a predisposing factor for stone formation [15]. |
| Anatomical abnormalities of the urinary tract | horseshoe kidney, ureteric stricture, pelvi-ureteric junction obstruction, ureterocele, caliceal diverticulum, medullary sponge kidney, and vesico-ureteric reflux [16]. |
| Gastrointestinal conditions | bariatric surgery, enteric hyperoxaluria, Crohn’s disease, jejuno-ileal bypass, and intestinal resection [17]. |
| Genetic conditions | primary hyperoxaluria, renal tubular acidosis, Dent’s disease, cystinuria, and cystic fibrosis [18]. |
| Conditions which alter urinary volume, pH, and/or concentrations of certain ions | hyperparathyroidism, nephrocalcinosis, diabetes, hypertension, polycystic kidney disease, gout, sarcoidosis, spinal cord injury, neurogenic bladder, and increased levels of vitamin D [19]. |
| Drugs | responsible for about 1% of all urinary tract stones [20]. |
KSD, kidney stone disease.
In assessing patients suspected of having KSD, the European Association of Urology (EAU) guidelines recommend a focused urological history, blood analysis (inflammatory markers, renal function tests, calcium and uric acid), urinalysis (+/- urine culture) and diagnostic imaging [21]. Most patients present with acute, severe loin to groin pain associated with nausea and vomiting (Fig. 1). Some will present with haematuria, while the majority will demonstrate microscopic haematuria on urinalysis.
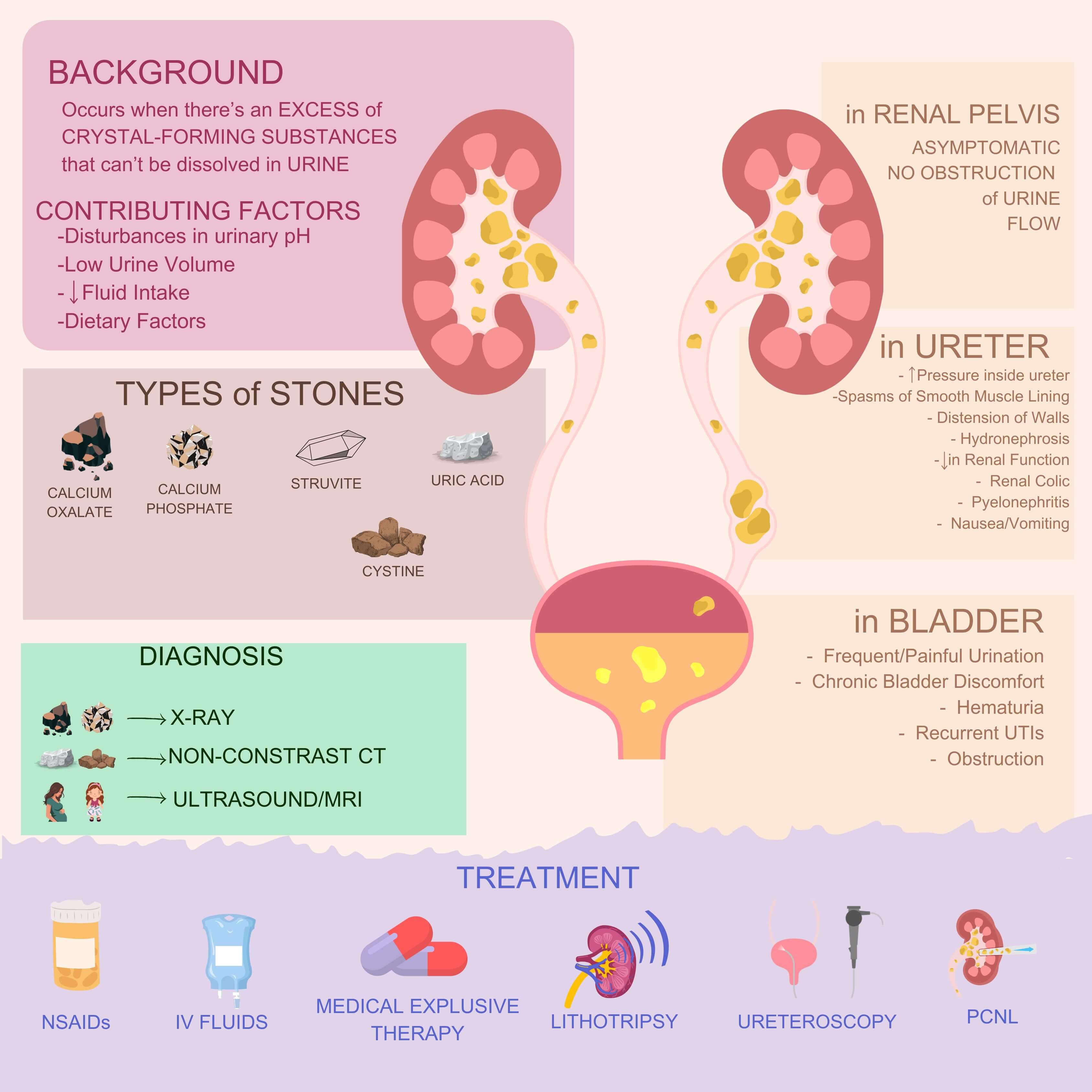 Fig. 1.
Fig. 1.
Clinical features, diagnosis and management of kidney stone disease (KSD). The figure was created with Canva Pro (version 1.104.0, Canva Pty Ltd., Sydney, Australia). CT, computed tomography; NSAIDs, non-steroidal anti-inflammatory drugs; PCNL, percutaneous nephrolithotomy; MRI, magnetic resonance imaging; IV, intra venous; UTIs, urinary tract infections.
Renal calculi can occasionally be discovered incidentally, during imaging conducted for the evaluation of haematuria or for other medical reasons. Symptoms are unlikely unless the calculi become infected or cause some degree of urinary tract obstruction, necessitating surgical decompression and drainage of the collecting system.
Urinalysis may demonstrate microscopic haematuria and urinary crystals which can be indicative of urolithiasis. Additionally, the presence of nitrites, leukocytes, and bacteria may suggest a urinary tract infection, requiring prompt culture and aggressive treatment.
Plain abdominal X-ray is particularly useful in differentiating between radiopaque and radiolucent stones, as well as in the context of follow-up evaluations.
Ultrasound is valuable for evaluating the presence of obstruction and the
subsequent development of hydronephrosis, particularly during pregnancy when the
use of X-ray imaging is generally avoided. Uric acid stones and other
non-calcific renal stones
Non-contrast computed tomography scan of kidneys, ureters and bladder (CT KUB) is the imaging gold standard for urolithiasis (Fig. 1). It can provide additional information regarding stone density, and skin to stone distance, which are pertinent considerations prior to extracorporeal shock wave lithotripsy (ESWL).
Metabolic evaluation of KSD aims to quantify the likelihood of urine crystallization, explore the metabolic pathways associated with urolithiasis, identify potential causes of this condition, evaluate the risk factors for chronic kidney disease (CKD) and mineral bone disease (MBD), and gain a deeper understanding of dietary habits.
The EAU guidelines recommend that all patients who present to hospital with a urinary tract stone should undergo a basic metabolic screen including urine dip stick, serum creatinine, Estimated Glomerular Filtration Rate (eGFR), sodium, potassium, ionised calcium and urate [21]. Likewise, stone samples retrieved from the patient are sent for biochemical analysis.
Patients at risk of KSD should aim for a healthy body weight (with a body mass index (BMI) range of 18–25 kg/m2), engage in sufficient physical activity and adhere to a nutritious and balanced diet characterized by a high vegetable and fibre intake, appropriate level of calcium consumption (1 to 1.2 g/day), low sodium chloride intake (4 to 5 g/day), and small amounts of animal protein content (0.8 to 1.0 g/kg/day) [11]. Specifically, consumption of low-fat dairy products and avoidance of highly processed foods, large amounts of red meat, spinach, rhubarb, beets, chocolates and nuts is recommended [22].
EAU guidelines have underpinned the beneficial role of ensuring a daily water
intake of
The citrate content of fresh fruit juice alongside bicarbonate plays an inhibitory role against calcium crystallization, that increases the alkalinity of urine. The presence of potassium further enhances both the pH levels and the concentration of citrate [23].
Reduced dietary calcium consumption should be discouraged for most patients, as it can enhance both the intestinal absorption and urinary excretion of oxalate, leading to an increased risk of stone formation [11]. It is recommended that individuals focus on limiting or avoiding foods that exhibit exceptionally high oxalate levels. A potentially effective approach to mitigating oxalate absorption involves the concurrent intake of oxalate- and calcium-rich foods [11].
Thiazide diuretics are known to lower urinary calcium excretion, which may improve bone mineral density [24] Therefore, it is recommended in patients with a history of calcium stone formation and calciuria.
Both allopurinol and febuxostat may be of benefit in patients exhibiting hyperuricosuria and forming calcium stones. Allopurinol plays a role in reducing urinary urate levels and lowering the recurrence of stones in individuals with hyperuricosuric calcium oxalate stones, provided they do not have other metabolic disorders [25].
Citrate use is recommended in patients with recurrent calcium oxalate (CaOx) nephrolithiasis characterized by reduced urinary citrate levels. Additionally, it is indicated in patients suffering from distal renal tubular acidosis, chronic diarrhoea, hypocitraturia and in stone formers with mineral bone disease (MBD). Potassium citrate is favoured over sodium citrate due to its role in reducing calciuria, and subsequently better prevention of calcium stone formation [8].
Adequate fluid consumption (
It is important to emphasize the importance of increasing fluid consumption to over 3 L/day, alongside the use of sodium bicarbonate or potassium citrate to increase urinary pH and enhance cystine solubility [21]. It is essential to monitor urinary pH multiple times throughout the day to appropriately adjust the dosage of the alkaline agents. The optimal pH range for effective management is between 7.0 and 8.0. In severe cases, the administration of 6-mercaptopropionyl glycine or d-penicillamine may be warranted [26]. As both pharmacological agents possess the potential to induce proteinuric glomerular diseases, regular urine assessments for proteinuria are recommended [21]. Patients diagnosed with cystine stones require rigorous monitoring due to the elevated metabolic activity associated with this condition.
Achieving complete struvite stone clearance is essential for effective prevention of recurrence. In fact, eradicating the infection becomes nearly unattainable if the stone remains, due to its role as a reservoir for microorganisms [21]. Following the extraction of the stones, a prolonged course of targeted antibiotic therapy may be required to maintain urine sterility and avert the formation of new stones.
Oral chemolysis is mostly effective in dissolving uric acid stones by adjusting urinary pH to between 7.0 and 7.2 using potassium citrate or sodium bicarbonate, increasing urine output and lowering uricosuria using allopurinol or febuxostat [27]. Although scarcely used these days owing to their potential toxicity, percutaneous irrigation solutions such as 10% hemiacidrin are effective for struvite, urate, cystine and brushite stones.
Adequate analgesia using primarily non-steroidal anti-inflammatory drugs (NSAIDs) (Fig. 2) and paracetamol is crucial in the immediate management of acute renal colic [21]. Physicians must bear in mind the risk of coronary events associated with diclofenac and ibuprofen use [28], as well as cardiovascular and upper gastrointestinal bleeding risks linked with long term use of diclofenac [29]. NSAIDs can impact renal function in individuals with pre-existing decreased eGFR. If pain relief cannot be achieved with medication, drainage using stents, percutaneous nephrostomy, or stone extraction, should be considered.
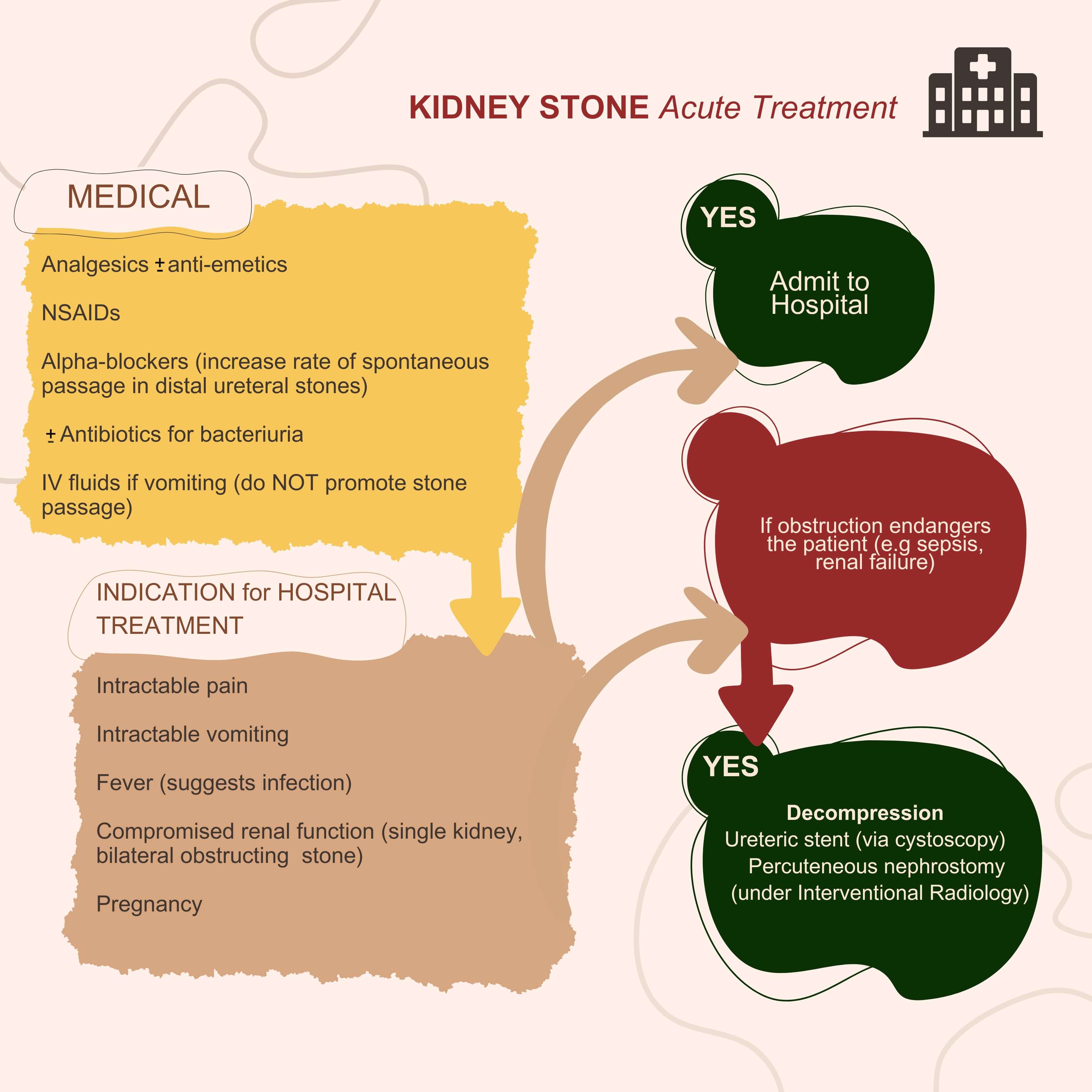 Fig. 2.
Fig. 2.
Acute treatment of KSD. The figure was created with Canva Pro (version 1.104.0, Canva Pty Ltd., Sydney, Australia).
Urosepsis and/or anuria in the setting of urinary tract obstruction is a urological emergency and should be managed by urgent decompression either by ureteric stent insertion or placement of a percutaneous nephrostomy tube. Whilst patients who undergo nephrostomy tube insertion tend to have a longer hospital stay, those who undergo ureteric stent insertion are more likely to be negatively impacted by stent intolerance [21]. A sepsis screen should be performed and targeted antibiotic therapy commenced immediately based on local policy, pending sensitivities [30].
The three most widely used methods for treating renal and ureteric stones are extracorporeal shock wave lithotripsy (ESWL), ureteroscopic stone fragmentation, and percutaneous nephrolithotomy (PCNL). The success rate of each modality will depend on the doctor’s experience, stone factors (size, location, density) and patient factors (body habitus, comorbidities, anatomy). An algorithm for deciding appropriate treatment modality is shown in Fig. 3.
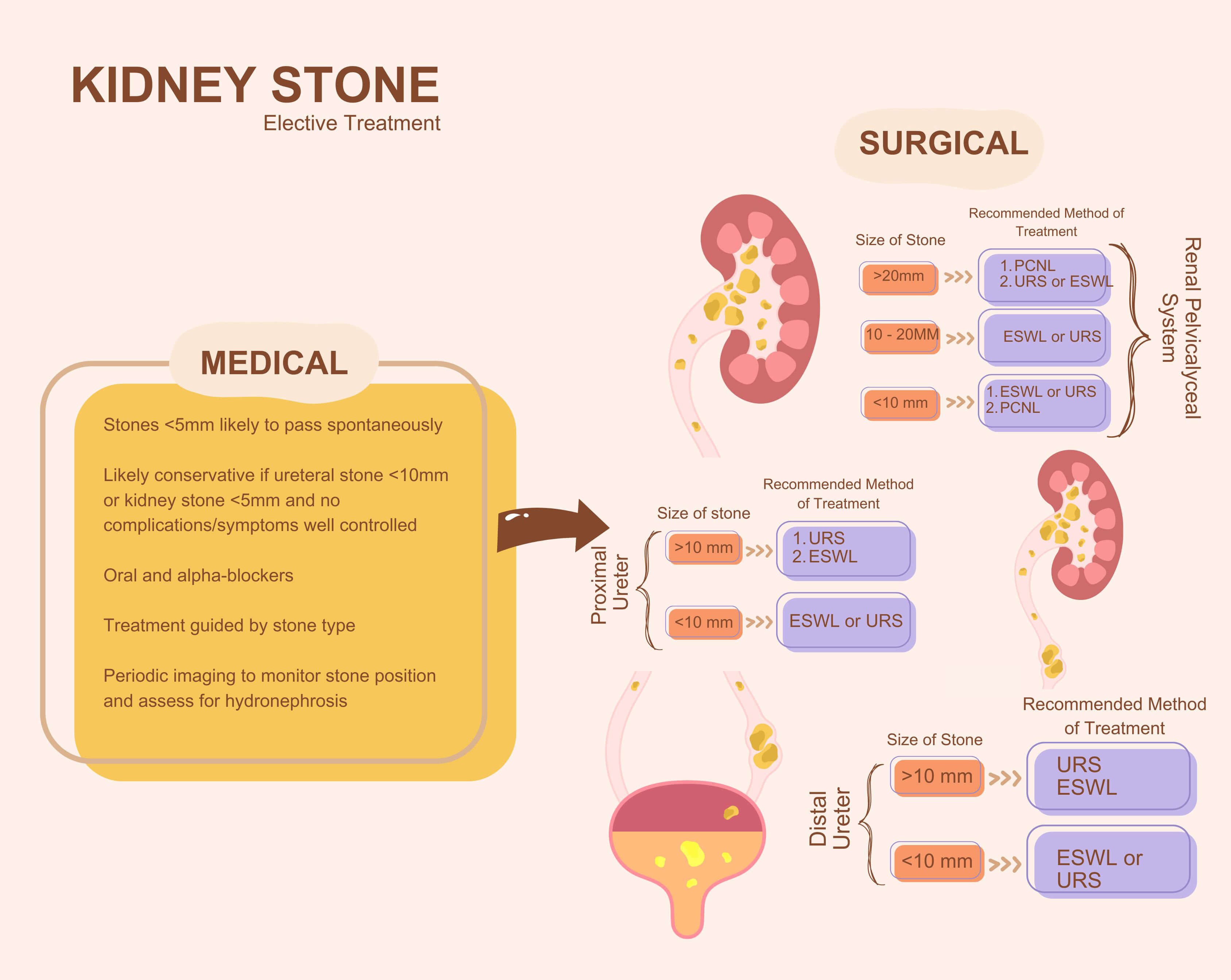 Fig. 3.
Fig. 3.
Elective treatment of KSD. The figure was created with Canva Pro (version 1.104.0, Canva Pty Ltd., Sydney, Australia). URS, ureteroscopy; ESWL, extracorporeal shock wave lithotripsy.
Shock wave lithotripsy (SWL) operates by generating high-energy shock waves using electrohydraulic, electromagnetic, or piezoelectric technology which release energy that breaks the stone. A specially designed focusing system ensures that the shockwaves are focused on the target stone, thus concentrating the energy at a specific point. Ultrasound or fluoroscopy ensures accurate location of the calculus, allowing precise targeting of the shock waves. Plain abdominal X-ray or ultrasound is usually used to assess the result of SWL within 3 months of the procedure.
Ureteroscopy involves the insertion of a thin telescope through the urethra and bladder, into the ureter and kidney. On gaining access to the stone, instruments like guidewires, laser fibres, balloon dilators, stone baskets and ureteric stents can be deployed. It is performed in the operating theatre under general or spinal anaesthesia. Whilst a semirigid ureteroscope is used for the distal ureter, a flexible ureteroscope is useful for accessing the proximal ureter and pelvicalyceal system. An access sheath can sometimes be used to ensure continuous flow, maintain low pressure, improve vision, and aid repeated stone extraction. Although the Holmium: yttrium-aluminum-garnet (Ho: YAG) laser is the more commonly used method of lithotripsy in most developed countries, the Thulium fibre laser (TFL) is now preferred in some centres due to its improved surgical outcomes [32].
Ureteroscopy has demonstrated better stone-free rates for kidney and ureteric
stones and requires fewer repeat treatments compared to SWL [33]. Therefore, for ureteral stones
PCNL involves passing an endoscope through the skin directly into the kidney. It is typically done for stones larger than 2 cm (Fig. 3). Under general anaesthesia, the patient is positioned either prone or supine and the kidney’s location is determined using anatomical landmarks. Renal access is then secured using a needle inserted under fluoroscopic and/or ultrasound guidance sometimes aided by retrograde studies. A guidewire is then inserted through the needle into the bladder and the tract dilated sequentially over a guidewire prior to placing a working sheath through which lithotripsy and stone extraction are performed using nephroscopes (rigid and flexible), a pneumatic and ultrasonic lithoclast and stone retrieval graspers.
Following the procedure, nephrostomy tubes may be left in place to facilitate optimal renal drainage. PCNL is said to be ‘tubeless’ when neither a catheter nor ureteral stent is left in place. Totally tubeless PCNL (without a ureteral stent or nephrostomy tube) is associated with shorter hospital stay, decreased analgesic requirements with no disadvantages reported [35].
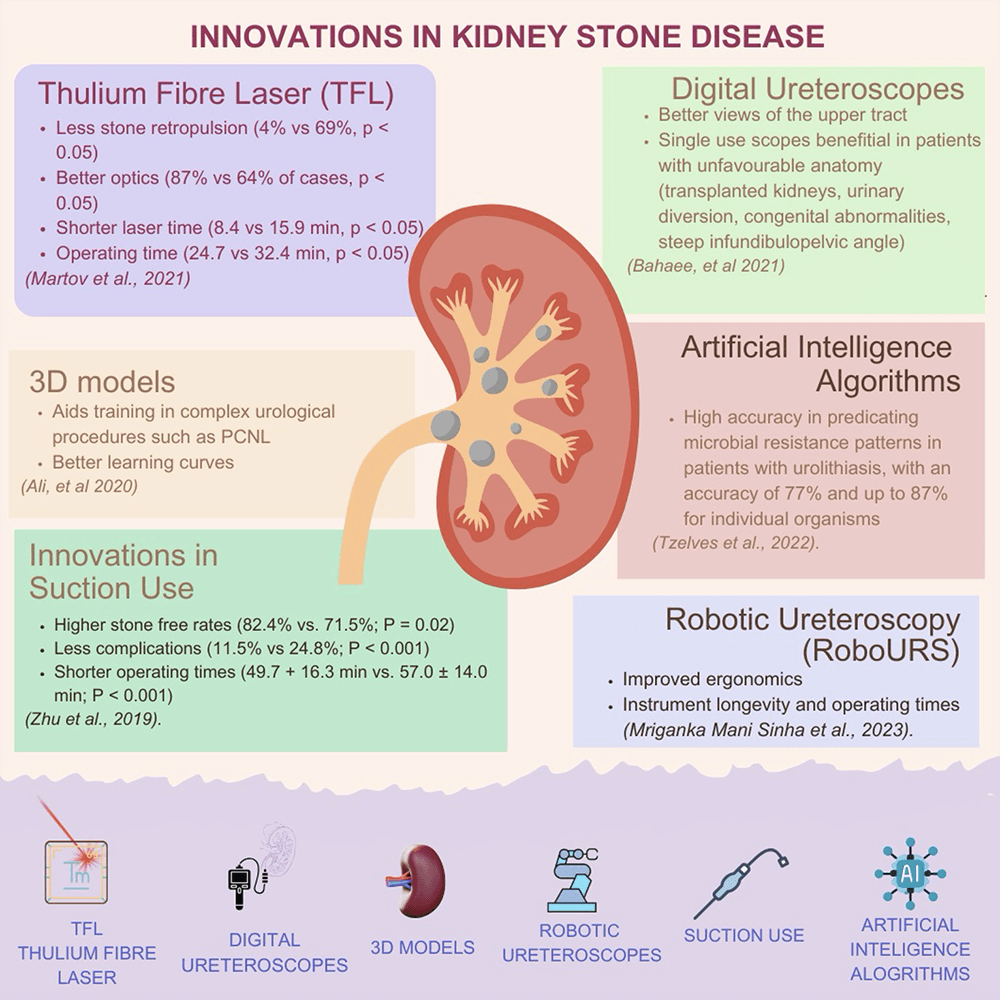
The past few years have seen advancements both in technique and technology used
in the management of urolithiasis (Fig. 4, Ref. [32, 36, 37, 38, 39, 40]). In some centres for example, there
has been a transition from the use of Holmium: yttrium-aluminum-garnet (Ho: YAG)
laser to TFL platform which has been shown to have less stone retropulsion (4%
vs. 69%, p
Patients who are stone-free may be discharged after two years for radiopaque stones or after three years for radiolucent stones, as research shows that 80% of them will remain stone-free in the long term [21]. To increase the safety margin and ensure that 90% remain stone-free, patients should be monitored for up to five years. However, those with metabolic abnormalities should undergo a more extensive follow-up [21].
The suggested imaging includes plain film X-ray of kidney, ureter and bladder (KUB) and/or ultrasound, depending on the type of stone and the preference of the clinician. A computed tomography (CT) scan should be used only for symptomatic disease or imaging before surgery, to prevent excessive radiation exposure [41].
The last three decades have witnessed a significant change in the management of KSD, with a shift to minimally invasive procedures becoming the norm. More recently, KSD management has benefited from technological advances with AI/3D models, virtual reality, robotics, advanced optics, digitalisation and miniaturisation featuring prominently not only in equipment design but also in several other aspects of clinical practice. Whilst these innovations have improved the safety and clinical outcomes of KSD management, research into the cost and quality of life implications should be considered.
All the data of this study are included in this article.
EO—Concept, manuscript writing; PJJ—Concept, manuscript editing; BKS—Concept, manuscript editing, supervision. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Figs. 1,2,3,4 were produced by Canva Pro. The authors confirm that every aspect of this manuscript has been rigorously reviewed and refined, and they take complete responsibility for its accuracy, integrity, and originality.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.