1 Guy’s and St Thomas’ NHS Foundation Trust, SE1 7EH London, UK
2 Royal Devon University Healthcare NHS Foundation Trust, EX2 5DW Exeter, UK
3 University of Exeter Medical School, EX1 2LU Exeter, UK
Abstract
Delivery of reperfusion therapies such as thrombolysis and mechanical thrombectomy have revolutionised the treatment for acute ischaemic stroke with reduction in disability and mortality. Extended time windows for thrombolysis for patients presenting beyond 4.5 hours including wake up stroke have now increased the eligibility for these treatments. Tenecteplase has emerged as alternative thrombolytic agent to alteplase, with practical advantages. Mechanical thrombectomy is now available to a wider group of patients than previously considered such as patients presenting beyond 6 hours up to 24 hours, those with large core anterior circulation infarcts and posterior circulation stroke. Advances in brain imaging and their application to determine salvageable brain tissue is crucial in delivery of these interventions. Systems of care to enhance the delivery of these time critical treatments such as pre-hospital video triage and mobile stroke units may play a vital role in maximising population benefits from high-quality hyper-acute stroke care. The purpose of this review is to highlight the expansion of evidence for acute therapeutic interventions for ischaemic stroke and technologies to facilitate their delivery.
Keywords
- ischaemic stroke
- acute
- thrombolysis
- thrombectomy
There has been considerable advancement in hyper-acute care for patients presenting with ischaemic stroke underpinned by considerable evidence base which has evolved over the last decade. The most recent evidence has been captured in the updated National Clinical Guideline for Stroke, published online at https://www.strokeguideline.org which provides comprehensive, evidence-based practice guidance to improve the quality of care to adults with a stroke in the United Kingdom and Ireland [1]. Advances in thrombolysis particularly in extended hours and in wake- up stroke have resulted in a higher number of patients eligible for such treatments. Alternative agents to alteplase such as tenecteplase may also present logistical advantages in its delivery. Mechanical thrombectomy remains the mainstay treatment to aid recanalisation of occluded large vessels and has been the principal development in reducing patient disability and mortality. Such advancement has seen its application in a wider spectrum of clinical presentations including extended time windows using advanced imaging as well as large core infarct volumes. Such interventions should be delivered as rapidly and efficiently as possible to maximise the population benefit within specialised organised stroke services in line with the ambitions of the National Health Service Long Term Plan [2]. The adoption of innovations such as mobile stroke unit and pre-hospital video triage may provide efficient mechanisms to facilitate rapid delivery of reperfusion therapies. This commentary reflects the current advancement in practice in reperfusion therapy for acute ischaemic stroke as well as systems of care to optimise the administration of such interventions. As the research evidence expands to a greater number of eligible patients, there is hope that a significant proportion of patients will endure less disability and improved survival after ischaemic stroke.
The advancement of acute reperfusion strategies for acute ischaemic stroke has
resulted in the need for immediate and time critical access for multi-modal
imaging such as computed tomography (CT) or Magnetic Resonance (MR) angiography
and perfusion when required. In order to maximise the delivery of reperfusion
treatments in a timely fashion, it is important that the sequence of imaging
required is carried in a single setting to avoid unnecessary delays [3]. Measurement of the 10-point Alberta
Stroke Program Early CT Score (ASPECTS) is a useful validated method of
quantifying early ischaemic changes [4]. The ASPECTS score
relates to the middle cerebral artery territory divided into 10 segments and
points are deducted for each area of early ischaemic change in the
supra-ganglionic and ganglionic levels. For patients being treated with
intravenous alteplase within 3 hours, an ASPECTS score
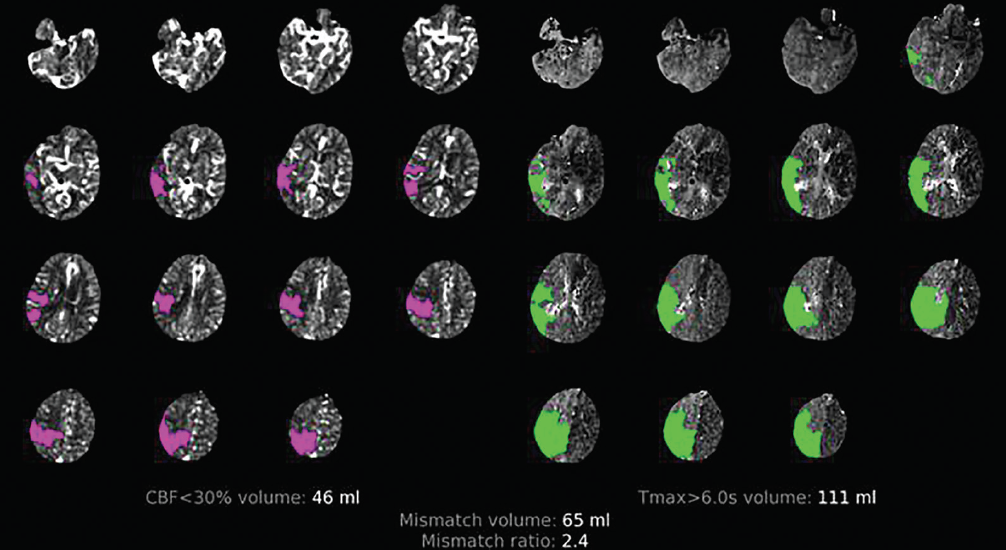 Fig. 1.
Fig. 1.
Computed tomography perfusion demonstrating cerebral blood flow
(CBF)
The evidence for intravenous thrombolysis with alteplase for patients with acute ischaemic stroke has progressed rapidly over the last few years. UK guideline recommendations for intravenous thrombolysis are no longer exclusively restricted to within 4.5 hours of known stroke onset [1]. The use of advanced imaging techniques such as CT perfusion and MR imaging have increased the eligibility of such patients to be treated up to 9 hours with alteplase after known stroke onset as well as ‘wake up stroke’ (9 hours from the midpoint of sleep) and patients with an unknown onset time, by identifying a radiologically defined ‘penumbra’ (potentially salvageable brain tissue) [8]. Quality improvement applications directed towards in-hospital metrics such as improving the door to needle time can help to augment the population benefit from thrombolysis and subsequent reduction in disability with the aspiration of improving national thrombolysis rates towards 18% [13]. Machine learning models can predict estimated achievable thrombolysis rate targets in hospitals through national audit, based on determining stroke onset times, applying a 30-minute door to needle time and adopting thrombolysis decisions made by the top 30 thrombolysing centres [13]. Thrombolysis with alteplase is currently administered to about 12% patients with acute stroke in the UK and Ireland but there are processes of care that can be made more efficient to significantly increase numbers who receive these interventions [14].
There is now compelling evidence that tenecteplase is a suitable alternative to alteplase as a thrombolysis agent in the management of ischaemic stroke, a recommendation subsequently supported by National Institute for Health and Care Excellence (NICE) [15]. Three recent randomised controlled trials have demonstrated non inferiority comparing tenecteplase (0.25 mg/kg) versus alteplase when delivered within 4.5 hours of stroke onset in relation to independent recovery and a study level meta-analysis has demonstrated superiority with regards to no disability at 90 days favouring tenecteplase [16, 17, 18, 19]. This equates to a number needed to treat of 33 for one additional patient with no disability compared to alteplase. There were no significant differences between the two agents in safety profile such as intracerebral haemorrhage or mortality. Tenecteplase is a genetically modifiable variant of alteplase and has greater fibrin sensitivity and therefore a potentially more powerful thrombolytic agent [20]. It also has a biologically longer half than alteplase and therefore has the advantage of being delivered as a single bolus agent [21]. Additional advantages over alteplase include the potential of reducing the risk of dosing errors, and rapid delivery of the bolus when considering inter-hospital transfers for mechanical thrombectomy [22] (See Table 1). Real world experience of the shift to tenecteplase use in North America and New Zealand have now confirmed feasibility with additional logistical advantages over alteplase such as faster door to needle times [23]. As the worldwide shortage of tenecteplase supply has now been addressed, it is expected that tenecteplase at a dose of 0.25 mg/kg will be available for use in the United Kingdom and Ireland to meet the anticipated clinical demand as the standard of care for intravenous thrombolysis. Whilst acknowledging that head-to-head comparisons between tenecteplase and alteplase have not been conducted in extended time windows (4.5 and 9 hours) and with wake up stroke, carrying out such trials to determine non inferiority or even superiority may be challenging due to lower incidence of these acute presentations. However, data from the TWIST study [11] comparing tenecteplase versus placebo in patients with wake up stroke using plain CT suggesting non-significant favourable outcomes as well favourable safety and efficacy data emerging from the Thrombolysis in Imaging Eligible, Late Window Patients to Assess the Efficacy and Safety of Tenecteplase trial (TIMELESS) and the Tenecteplase Reperfusion Therapy in Acute Ischaemic Cerebrovascular Events-III trial (TRACE 3) (see below), this has been used to support the use of tenecteplase in these scenarios and have been recommended in UK consensus statements to simplify treatment pathways [24].
| Alteplase | Tenecteplase | |
| Composition | Recombinant DNA version of tPA | Genetically modified form of recombinant human tPA |
| Biological half life | 4–6 minutes | 20–25 minutes |
| Fibrin specificity | Lower fibrin affinity than tenecteplase | 15-fold higher fibrin specificity than alteplase |
| Risk of angioedema | 1–5% | 0.2 to 2.8% |
| Symptomatic intracranial haemorrhage | 3.6% | 1.8% |
| Delivery | Bolus 10% administered intravenously over 1 minute, with remaining dose delivered as an infusion over 60 minutes | 5-second intravenous bolus |
| FDA approval for ischaemic stroke | Yes | Off label use |
tPA, tissue plasminogen activator; FDA, Food and Drug Administration.
Some caution may need to be applied to patients with non-disabling stroke with intracranial occlusion treated up to 12 hours after onset as the Tenecteplase Versus Standard of Care for Minor Ischaemic Stroke with Proven Occlusion (TEMPO-2) trial suggested no benefit and possibly harm (increase in symptomatic haemorrhage and death) with tenecteplase, however further advanced imaging may determine a subgroup of patients who may be a lower risk of intracerebral haemorrhage [25]. At present, patients with minor stroke (National Institutes for Health Stroke Scale (NIHSS): less than 5) should still be or consideration for treatment with intravenous thrombolysis if the deficits are thought to be disabling to the patient. There is also evidence that superior recanalisation occurs in patients with large vessel occlusion treated with tenecteplase compared to alteplase within 4.5 hours of stroke onset followed by thrombectomy as demonstrated in the Tenecteplase versus Alteplase before Endovascular Therapy for Ischemic Stroke trial (EXTEND-IA TNK), with improvement in disability as a secondary outcome [26]. However, real world data from multicentre registries have not been confirmatory for greater early recanalisation and subsequent subgroup analysis of randomised controlled involving patients with large vessel occlusion have been equivocal [22].
The use of tenecteplase in treating patients with large vessel occlusion in the extended time window (between 4.5–24 hours) using advanced perfusion imaging in determining salvageable brain tissue has also been evaluated in two studies [27, 28]. The TIMELESS trial, demonstrated no significant difference in functional outcome in patients treated with tenecteplase compared with initial standard care followed by thrombectomy in patients who were managed in comprehensive stroke centres, however tenecteplase was deemed to be safe with no difference in mortality or symptomatic intracranial haemorrhage (3.2% tenecteplase vs 2.3% standard treatment) at 90 days [27]. One possible explanation of the neutral results may relate to the evidence that the benefits of thrombolysis in combination thrombectomy is observed particularly if thrombolysis is delivered rapidly (at least within 140 minutes of stroke onset) for patients presenting directly comprehensive stroke centres [29]. In addition to this, most patients were treated with thrombectomy shortly after receiving intravenous tenecteplase (a median of 16 minutes). In contrast, TRACE 3 study demonstrated that tenecteplase was beneficial in enhancing functional outcome compared to patients receiving standard care in the extended time window in patients where thrombectomy was not undertaken. Despite the improvement in recanalisation and early recovery, the rate of symptomatic intracranial haemorrhage was higher in the tenecteplase arm (3%) versus 0.8% in the control arm. These results may suggest a use for tenecteplase when considering inter-hospital transfer for possible thrombectomy [28].
Mechanical thrombectomy is currently the most effective reperfusion therapy in
reducing stroke related disability in patients with large vessel occlusion (the
distal internal carotid artery, or the first or proximal second divisions of the
middle cerebral artery). Individual patient meta-analysis of five landmark trials
(MR CLEAN [30]; EXTEND-IA [31]; ESCAPE [32]; REVASCAT [33]; SWIFT PRIME [34]) evaluating mechanical thrombectomy delivered within 6 (and up to a maximum of 12) hours after onset in patients with large vessel occlusion in the anterior circulation, demonstrated that the numbers needed to treat for one additional patient to have reduced disability by one point on the modified Rankin Score (mRS) at 90 days was 3 [35]. Patients included had a measurable neurological deficit and were relatively independent prior to their stroke (mRS
There is increasing evidence that patients also benefit from mechanical thrombectomy delivered in the extended time window (between 6–24 hours after onset) using advance imaging such as CT and MR perfusion involving the anterior circulation [37, 38]. Identification of perfusion or clinical core mismatch has been used as a means of patient selection between 6 and 16 hours and 12–24 hours with improvement in functional outcome, acknowledging however that a significant population of patients presenting late during these extended time windows would still be ineligible [37, 38]. These criteria have now been superseded with increasing evidence underpinning the delivery of mechanical thrombectomy in patients with large core infarcts up to 24 hours who had previously been ineligible for earlier trials beyond 6 hours [9, 10]. Imaging criteria included the ASPECTS score based on plain CT imaging, CT angiography to identify large vessel occlusion or advanced imaging to evaluate perfusion mismatch and core volume. The randomised controlled trial to optimise patient’s Selection for Endovascular Treatment in Acute Ischemic Stroke (SELECT 2) demonstrated that mechanical thrombectomy resulted in improved functional outcome (mRS 0–2) by 13% (Numbers needed to treat: NNT = 8) at three months compared to standard treatment in patients with large core infarcts (ASPECTS score 3–5) and internal carotid artery and proximal middle cerebral artery occlusions treated up to 24 hours [10].
ANGEL-ASPECT study also demonstrated improved functional recovery by 19% (NNT = 6) at three months despite higher rates of intracranial haemorrhage in patients with large core volumes (ASPECTS score 3–5) with core volumes (70–100 mL) up to 24 hours of onset. However, the median mRS was 4 indicating that a significant proportion of patients will still be disabled and require on going specialist multidisciplinary rehabilitation [9]. Decision making to intervene for patients with large core infarcts should take account of the patient’s pre-stroke dependency, underlying frailty and patient wishes. Current guidance supports the use of mechanical thrombectomy in patients presenting with acute anterior circulation ischaemic stroke and large artery occlusion between 6 and 24 hours of onset, including wake-up stroke on the basis of a combination of ASPECTS score and clinical imaging mismatch using CT or MR if the following criteria are satisfied, irrespective of core infarct size [1]:
(1) Between 6 and 12 hours: an ASPECTS score
(2) Between 12 and 24 hours: an ASPECTS score
Benefits in improved recovery, quality of life and survival were also observed
in patients with large core infarcts treated by mechanical thrombectomy within 12
hours of stroke onset longer term up to 12 months using CT and CT angiography
(CTA) in the TENSION study [39]. However, it should be
acknowledged that in this study, the median mRS was 5, indicating high levels of
disability in the treatment arm. A subsequent meta-analysis of large core
infarcts including SELECT 2 [10], ANGEL-ASPECT [9], RESCUE Japan [40], TESLA [41], TENSION [39] and LASTE [42]
treated with mechanical thrombectomy up to 24 hours with ASPECTS scores
There have been two trials from China showing benefit with mechanical thrombectomy versus best medical treatment within 24 hours for patient with basilar artery occlusion with moderate to severe symptoms and without evidence of large-scale established infarction in the posterior circulation at presentation. The ATTENTION trial demonstrated an absolute difference in mRS 0–3 of 23.2% favouring mechanical thrombectomy versus best medical therapy alone in a time window of up to 12 hours after stroke onset [46]. The BAOCHE trial also showed superiority for thrombectomy with an absolute difference in mRS 0–3 of 22.1% in patients treated between 6 and 24 hours, although there is still paucity of data for patients presenting beyond 12 hours, aged over 80 years old and patients with mild symptoms [47].
There are still a number of knowledge gaps in our understanding of which patients may benefit from mechanical thrombectomy. These include identifying which patients with large core infarcts benefit including patients with frailty, multi-morbidity and those older than 80 years of age [48]. Trials have focussed on intracranial large vessel occlusion presentations but recent studies have demonstrated no significant benefit in functional recovery and higher rates of symptomatic intracranial haemorrhage and mortality with mechanical thrombectomy involving distal medium sized vessels in addition to thrombolysis [49, 50]. Patients with mild stroke impairments which may be still debilitating to the patient have also been underrepresented in trials and require further exploration. Adjuvant therapies such as interventions to enhance microvascular occlusion and cyto-protection (freezing the ischaemic penumbra) may be complimentary to mechanical thrombectomy to aid reperfusion [51].
There is a need for health services to support the delivery of thrombectomy to an increasing eligible population through extending 24/7 coverage amongst thrombectomy centres, access to artificial intelligence for brain imaging to aid decision making, workforce capacity and efficient pre-hospital and inter-hospital pathways in a networked approach. Current rates of thrombectomy are just over 4% and estimates suggest that these recommendations as well as wider use of advanced imaging selection will extend eligibility for thrombectomy to 15% of hospitalised stroke [52].
Pre-hospital emergency services should be organised locally and regionally to ensure the delivery of thrombolysis and mechanical thrombectomy to the local population [53]. Whether patients should bypass their local acute centre to be directly transferred to a thrombectomy centre is unclear. The RACECAT trial in Spain did not demonstrate an outcome advantage for patients with suspected large vessel occlusion who were directly transferred to a thrombectomy centre compared to their local acute stroke centre, however outcomes for patients with intracerebral haemorrhage were worse for those patients who bypassed their local stroke centre [54]. Current research is in progress in the UK exploring pre-hospital assessments whereby patients who may be considered for mechanical thrombectomy bypass their local acute stroke centre and are directed to a comprehensive stroke centre [55]. Prehospital video triage has provided a vehicle to allow hospital-based stroke specialists in adopting digital videoconferencing to communicate with ambulance clinicians at the scene of suspected stroke patients [56, 57]. This modality has improved conveyance of suspected stroke patients to hyper-acute stroke services, improved time metrics for thrombolysis and thrombectomy as well as redirection of stroke mimics to non stroke emergency services [58]. Although favourable effects on usability, acceptability and patient safety have been reported, its implementation relies on robust governance, training, as well as front line clinicians and paramedics that are receptive working within a network structure [56, 57, 59]. Further research is required to evaluate their cost effectiveness, implementation and user experience through the PHOTONIC study [56]. Mobile stroke units (MSUs) are ambulances assembled with CT-scanning equipment and specialist staff that are capable of delivering thrombolysis at the scene and/or identifying large artery occlusion when equipped with CT angiography. Non randomised trials evaluating mobile stroke units have shown improved clinical outcomes, higher rates of delivery of thrombolysis and faster times from onset to thrombolysis compared with standard ambulance practice involving sites in Germany and North America, however whether these services can be deployed in metropolitan or urban areas in the UK amongst an unselected population is unknown and requires further cost evaluation [60]. There is some evidence that MSUs may be cost effective during the first year in delivering thrombolysis in patients without pre-existing disability compared to standard emergency care services [61].
New recommendations for thrombolysis and thrombectomy will be key to improve the quality of care and reduce disability and dependency for stroke patients. The challenge ahead lies with implementing the recent updated guidelines and organisations that are responsible for planning and delivering stroke services should ensure these recommendations are fully supported in an ever-changing and highly pressured healthcare landscape. Clinical networks play a crucial role in collaborating with providers and commissioners to enable innovative systems of care to enhance the delivery of reperfusion therapies as rapidly as possible, such as artificial intelligence use for advanced imaging, pre-hospital video triage and mobile stroke units. Using comprehensive, prospective national stroke audit data through the Sentinel Stroke National Audit Programme to explore the pre-hospital and hyper-acute pathway will provide a vehicle for quality improvement to transform stroke care for the future.
All the data of this study are included in this article.
AB and MJ were contributors in the conception and writing of this review. AB drafted the manuscript. Both authors contributed to important editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

