1 School of Healthcare Sciences, Cardiff University, CF14 4XN Cardiff, UK
Abstract
This review presents an outline of the state of the knowledge on the prevention and diagnosis of cervical cancers over the last decade. Screening and prevention methods are considered, such as cervical self-sampling, Human Papillomavirus (HPV) vaccination and the use of digital technologies and artificial intelligence (AI) in diagnostics and screening, as are some success stories. However, despite the successes stemming from the UK and Europe–such as that no new cases of cervical cancer have been detected in a cohort of Scottish women vaccinated against HPV between 12–13 years old–cervical screening rates are falling and social, cultural and economic barriers to HPV vaccination uptake remain. There are several clear pathways that could reduce not only incidence rates of cervical cancer, but also non-cervical HPV related cancers such as oropharyngeal and anal cancers.
Keywords
- Papillomavirus infections
- Papillomavirus vaccines
- uterine cervical neoplasms
- oropharyngeal neoplasms
- artificial intelligence
- Europe
- UK
This review will assess progress in the prevention and diagnosis of cervical cancers over the last decade, specifically focusing on the UK and Europe, but with reference to other areas where relevant. The current data surrounding cervical cancer prevalence is provided, as is the current state of screening and prevention methods. A number of success stories are also provided. The focus then moves on to explore the future challenges and pathways forward. Regarding Human Papillomavirus (HPV) vaccination, disparities between European regions are addressed, as are the reasons surrounding vaccine hesitancy in areas such as Eastern Europe. These barriers to uptake sit across social, cultural and economic factors. One of the ways forward could be in targeted educational interventions, alongside gender neutral vaccination and innovative digital tools. Regarding cervical cancer screening, barriers to attending appointments exist. In order to combat these barriers, self-sampling is being trialled, as are self-testing devices using menstrual blood and urine, alongside electronic health interventions. Finally, new diagnostic methods and the use of artificial intelligence (AI) are examined. The review ends by considering the rising incidence rates of non-cervical HPV cancers, particularly types that affect men such as oropharyngeal, anal and penile cancer, reinforcing the need for gender neutral vaccination worldwide.
Cervical cancer occurs when abnormal cells grow uncontrollably in the lining of
the cervix, eventually forming a tumour. Common symptoms include unusual vaginal
bleeding, pain during sex or vaginal discharge, although not everyone will
experience symptoms [1]. Survival rates vary
depending on the stage of cancer at diagnosis but generally, one-year survival is
The latest data shows that, in the UK, cervical cancer is now the 14th most common cancer in females, with around 3300 new cases each year, equating to around nine cases per day [4]. It is most common in females aged 30–34 years old. Although incidence rates have remained stable over the last decade, in England, incidence rates are 65% higher in the most deprived quintile of the population. Overall, it is thought that 99.8% of cases are preventable through methods such as cervical cancer screening and HPV vaccination.
The UK’s National Health Service (NHS) first began offering cervical cancer screening in 1964. The programme is targeted at females aged 25–64 years, in addition to anyone with a cervix within this age range (e.g., transgender men, non-binary assigned female at birth). Screening is offered every 3–5 years (dependent on nation) [5]. Screening tests for the presence of HPV, which can cause cervical cells to become abnormal and is spread through close skin-to-skin contact, typically during sexual activity. HPV is common and for most people does not cause any harm. A free of charge vaccine is currently available through the NHS to all children 11–13 years old, people who missed their vaccine (up to 25 years old), men who have sex with men (up to 45 years old), and some transgender people (up to 45 years old) [6]. Gender neutral vaccination is promoted in the UK to increase herd protection and address equality.
Table 1 (Ref. [7]) highlights changes in vaccination uptake over the last decade by World Health Organization (WHO) regions, with European data showing that vaccination in females has increased by 14% since 2013, and 20% for males. Regions that have not seen increases in male vaccine uptake include Africa (with only a 1% increase), the Eastern Mediterranean (with a 0% increase), South-East Asia (with a 0% increase) and Western Pacific (with a 1% increase). The Americas have seen success in male vaccination rates, increasing by 30%. Countries with significant increases in female vaccination rates include Africa (31% increase) and the Americas (26% increase) [7]. Additionally, Fig. 1 (Ref. [7]) demonstrates the increase in vaccination coverage in female and male cohorts across the European region over the last decade.
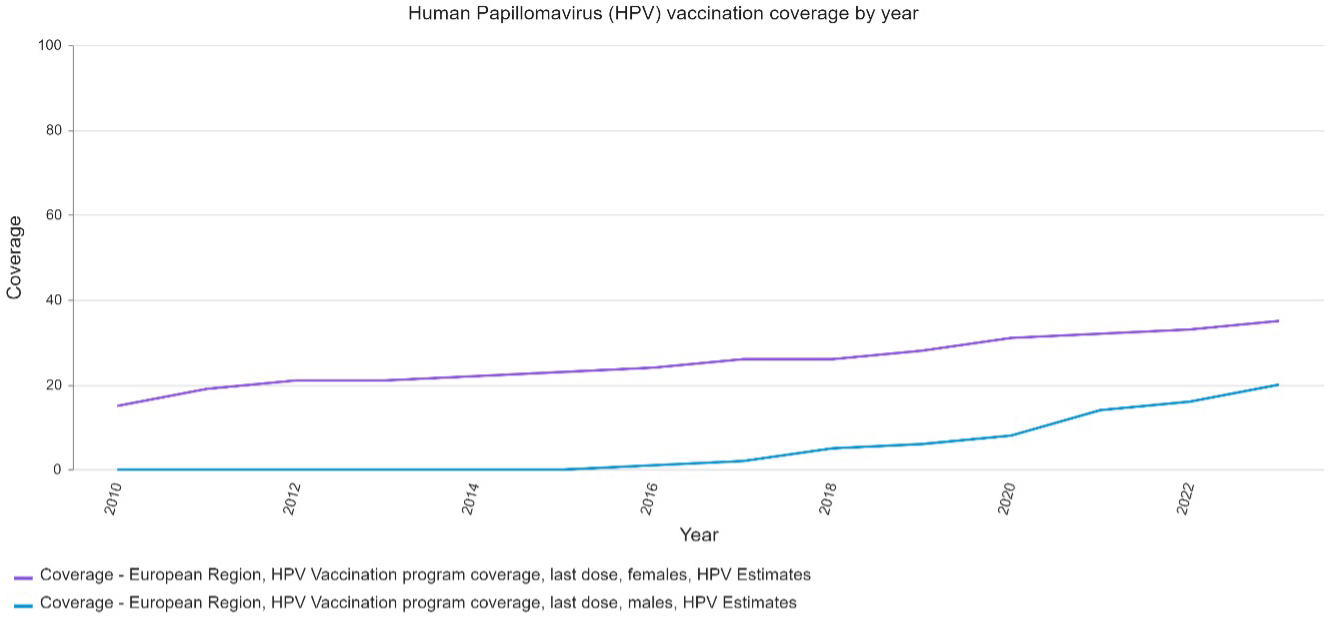 Fig. 1.
Fig. 1.
HPV vaccination coverage across the European region by year. The figure was created from WHO Immunization Data portal, [7].
| 2013 | 2023 | |||
| WHO Region | HPV Vaccination Programme—last dose (females) | HPV Vaccination Programme—last dose (males) | HPV Vaccination Programme—last dose (females) | HPV Vaccination Programme—last dose (males) |
| African | 1% | 0% | 32% | 1% |
| Eastern Mediterranean | 0% | 0% | 2% | 0% |
| European | 21% | 0% | 35% | 20% |
| The Americas | 29% | 3% | 55% | 33% |
| South-East Asia | 0% | 0% | 8% | 0% |
| Western Pacific | 4% | 0% | 7% | 1% |
The table is adapted from WHO Immunization Data portal, [7], available under the CC BY-NC-SA 3.0 IGO (https://creativecommons.org/licenses/by-nc-sa/3.0/igo). HPV, Human Papillomavirus; WHO, World Health Organization.
The WHO has set a target to eliminate cervical cancer by 2046 under their Cervical Cancer Elimination Initiative. This will involve fully vaccinating 90% of girls by the time they are 15 years old, ensuring that 70% of women are screened initially by 35 years–and again by 45 years–and effectively treating and/or managing 90% of women with cervical cancer disease (both pre-cancer and invasive cancer). If these targets can be met by 2030, more than 74 million new cases of cervical cancer will be prevented worldwide [8]. There are currently several countries on their way to achieving this–one being the UK. One estimate of the cost savings from achieving the WHO target in the UK sits at £2.6 billion [9].
The UK is on track to meet the WHO’s elimination targets by 2046 [9], but NHS England have recently stated that they aim to eliminate cervical cancer six years earlier by 2040 [10]. Initiatives such as self-sampling (currently being trialled) and gender neutral HPV vaccination programmes may assist in reaching the elimination targets.
There are a number of success stories across the UK, for example, a 2021 study demonstrated that cervical cancer is almost eliminated in England in females born since 1 September 1995, with the incidence decreasing by 87% for those women who were vaccinated when they were 12–13 years old [11]. The vaccination programme has been hugely successful, with around 80% of teenagers having already been vaccinated against Human Papillomavirus (HPV). The success is partly attributable to the ease of getting vaccinated, as the vaccine is offered in schools, free of charge [12].
Additionally, Scotland was the first of the four UK nations to introduce a
school-based vaccination programme, starting in 2008 [13]. No new
cases have been detected in any females born between 1 January 1988 and 5 June
1996, who received the vaccine at 12 or 13 years old [14].
The UK has also been given an HPV Prevention Policy Atlas ranking index of 95.8%
based on HPV awareness, which is measured by primary and secondary prevention,
and availability of online information. Other European countries with rankings
A report published by the European Cancer Organization (ECO) highlights national campaigns in the EU (Ireland, Greece and The Netherlands) that have either been successful or appear to be promising [16]. These include policy initiatives, catch-up programmes, awareness campaigns and educational activities to challenge misinformation.
A number of key policies have been designed in this area, such as the Recommendation on Vaccine-preventable Cancers [17], the European Region Roadmap [18], the aforementioned Cervical Cancer Elimination Initiative [8], and Europe’s Beating Cancer Plan [19]. The Beating Cancer Plan, launched in 2021, has offered some positive impacts such as through the imaging initiative and inequalities register [20], alongside agreed milestones surrounding progressive legislation and policy action [21]. The next steps are to refresh and expand the plan [22].
Key programmes tackling HPV vaccination include the ECO’s Protect Europe project. Co-funded by the European Union, it was launched in 2023 and ran for two years, aiming to increase vaccination in young people across Europe. As an excellent example of how to impact uptake, European member states have received guidance on how healthcare workers can best communicate with young people and their guardians, delivering effective public health messaging, and other relevant innovations [22].
Despite a significant amount of progress over the last decade and several success stories now emerging, challenges do remain that need to be addressed as we move forward. Across Europe, there are wide ranging disparities, with a 2020 study finding that incidence rates of cervical cancer range from 6.8 per 100,000 women in Western Europe compared to 16 per 100,000 in Central and Eastern Europe [23]. For example, in Bulgaria, the incidence per 100,000 women is 28.2, compared to in the UK where the rate is 11. This is especially significant given the difference in population size (3,600,000 in Bulgaria vs. 34,180,000 in the UK) [24, 25]. By the end of 2022, vaccination was offered in 42 out of 53 EU member states (79%), including in 31 out of 33 high-income countries (94%) and 11 out of 20 middle-income countries (55%) [26]. In early 2025 the World Health Organization (WHO) provided updated figures, reporting that 47 out of 53 countries now offer vaccination (89%), demonstrating that progress is being made [27]. However, there have been consistently low uptake rates in Eastern European countries, such as Romania and Bulgaria, with only 6% of females fully vaccinated in both countries [7] and with the incidence of cervical cancer in Romania being 2.5 times higher than the European average [19]. This is thought to be due to vaccine hesitancy and issues surrounding poor access [22]. Barriers to HPV vaccination have been well documented, with research showing that social, cultural and economic factors all contribute to low uptake, including religious beliefs, perceived links to promiscuity, the cost of the vaccine, mistrust of government and pharmaceutical agencies, worries about safety and side effects, as well as a lack of appropriate information being available [28, 29]. Meanwhile, flawed approaches exist in some European countries, such as in the case of Ukraine, where the vaccine is recommended but there is no national immunisation programme [15].
When considering how to tackle barriers to uptake, the effectiveness of interventions needs to be considered. For example, Bennett et al. [30] found that educational interventions are more effective when targeted at children, adolescents and parents, rather than parents alone. We also know that interventions must work across all levels of the health system. Socio-ecological model can help in this process. For example, Bronfenbrenner’s [31] socio-ecological model offers five levels that encompass influences from family, peers, school, community, the media and government [16]—these include the microsystem, mesosystem, exosystem, macrosystem and the chronosystem. By considering these different levels of influence, the complex challenges associated with the promotion of HPV vaccination can be better understood, and more importantly, addressed.
Furthermore, gender neutral vaccination campaigns are key. However, recent literature has identified that barriers exist, such as that boys have lower levels of knowledge about HPV infection and vaccination in comparison to girls and that there are misconceptions surrounding HPV being perceived as a women’s only problem [32]. Thus, targeted educational interventions are needed. Finally, digital tools such as mobile phone applications and websites have been found to be successful in increasing vaccine confidence and achieving user acceptability [33, 34, 35].
A need also exists to increase rates of cervical cancer screening by reducing barriers to appointment attendance and by offering alternative methods of screening. Despite high screening rates, attendance in the UK is decreasing compared to previous years [36, 37], with around one third of eligible women not attending screening appointments [38]. Research shows that women often have a negative perception of screening due to psychological and practical concerns, such as fear, embarrassment, worries about pain, inconvenient appointment times and locations, and anxiety surrounding having a male nurse. Pop-up clinics have been suggested by research participants who perceived General Practice (GP) surgeries to be inaccessible, particularly during working hours, alongside negative personal accounts found on social media [37, 39].
As one way to combat these barriers, the WHO has recommended that self-sampling be made available to women aged 30 and above [40]. Trials exploring this in England include ‘YouScreen’ and ‘HPValidate’, with the trials finding that self-sampling could be ‘game changing’ in improving screening numbers and that participants’ experience of self-sampling was excellent [41, 42]. The ‘YouScreen’ trial resulted in a 22% increase in non-attenders screened each month [41] and the ‘HPValidate’ study identified four combinations of self-collection devices and tests that worked well together and have been recommended for further validation [43]. One study currently running in Swansea, Wales, aims to explore patients’ recommendations and observations on how the service should be rolled out and what communications would be valuable to patients in facilitating decision-making [44].
Electronic health interventions offer additional methods for increasing screening attendance rates, with research finding that messaging services, telephone calls, online videos and internet booking increase screening rates, particularly among women in low- and middle-income areas [45]. Services such as these are being explored in the UK, with NHS England introducing a ‘ping and book’ service that will make booking screening appointments easier [46].
Alongside reducing barriers to attendance of cervical screening appointments and introducing self-sampling, there have been a number of recent advancements that offer innovative methods of diagnosing cervical cancer. These include DNA methylation triage, HPV integration detection, liquid biopsies and the use of artificial intelligence (AI) [47]. While the cervical ‘pap’ or ‘smear test’ has been the gold standard for many years and commonly used in healthcare systems such as the NHS, evidence now shows that HPV DNA-based tests are more accurate than cytology-based tests–especially in low- and middle-income countries–with this now being the screening method recommended by the World Health Organization [48].
Some UK-based innovations include ‘Papcup’, a self-testing device that looks for HPV strains in menstrual blood [49] and ‘Alternative Cervical Screening’ (ACES) that offers screening using urine samples [50]. The UK Government have also recommended the use of digital pathology techniques during cervical (and other cancer) screening [51]. AI screening systems are also being developed, trialled and implemented worldwide, especially in relation to grading in cervical cytology, as well as in colposcopy, magnetic resonance imaging (MRI) diagnosis and tumour staging [52, 53, 54].
Furthermore, HPV can cause other types of cancer including anal, penile, vulval, vaginal and oropharyngeal (a type of head and neck cancer). In fact, authors [55, 56] have found that HPV infection is associated with around 88% of anal, 50% of penile, 25% of vulval, 78% of vaginal and 31% of oropharyngeal cancer cases globally. Regarding penile and anal cancers specifically, the Global Cancer Observatory prediction tool estimates that cases will increase by around 78% and 72% respectively between 2022–2050 [57, 58]. Despite this, cervical cancer remains the only HPV-associated cancer that is screened for at present.
Cases of oropharyngeal cancer linked to oral HPV infection are on the rise, with incidence of HPV related oropharyngeal cancers surpassing cervical cancer in countries such as the USA and UK [59, 60]. Rates are rising, particularly in male cohorts across Northern Europe, the UK and North America [59, 61]. For example, in England alone, there was an overall 47% increase in cases of oropharyngeal cancer between 2013–2019, with males having over double the incidence rate than females [62]. This, alongside the high percentage of anal and penile cancer cases associated with HPV infection, reinforces the need for gender neutral vaccination campaigns. Educational interventions targeted at young men are also crucial as this group is typically more likely to present with HPV related oropharyngeal cancers [63].
Over the last ten years, there have been a number of developments concerning the prevention and diagnosis of cervical cancer, alongside many success stories. Regarding prevention, vaccine uptake has increased across Europe, with some countries now offering gender neutral vaccination programmes. Elimination targets set by the WHO are anticipated to be achieved by the UK some six years early, with the UK and other European countries also receiving higher rankings by the HPV Prevention Policy Atlas. However, disparities across Europe remain, with Central and Eastern Europe falling behind due to access issues (e.g., lack of national vaccination or screening programmes and/or vaccines not being free) as well as vaccine hesitancy. Gender neutral campaigns are also not yet the norm across the region.
Relating to screening and diagnosis, despite free cervical cancer screening in the UK, attendance rates are falling. To combat this, self-sampling methods are being trialled as a means of by-passing some of the barriers that prevent women from booking and attending appointments, such as inconvenience and fear. Self-sampling has also been recommended on a wider scale by the WHO. Finally, non-cervical HPV cancers require more thought and attention, including anal, penile, vulval, vaginal and oropharyngeal cancers which are often associated with HPV infection but are not screened for. Gender neutral vaccination campaigns are once again crucial in reducing incidence of several of these HPV cancers, as is reducing the societal barriers to vaccination. Overall, while progress and learning have been achieved, several challenges remain. The next steps required to tackle these challenges include ensuring national cancer plans set detailed targets for HPV vaccination uptake, increasing understanding of reasons for vaccine avoidance and tackling avoidance using up-to-date, evidence-based information, and finally, addressing variance across Europe to reduce the incidence and costs (both human and financial) of cervical and other HPV cancers.
All data generated or analysed during this study are included in this published article.
HW and DK were contributors to the conception and analysis of this review. HW and DK drafted the manuscript. Both authors contributed to the editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

