1 Department of Medicine, Medicine Academic Programme, SingHealth, 168582 Singapore, Singapore
2 Department of Medicine, Duke-NUS Medical School, 169857 Singapore, Singapore
3 Department of Renal Medicine, Woodlands Health, 737628 Singapore, Singapore
4 Monash Rural Health Mildura, Monash University, Mildura, VIC 3500, Australia
5 Department of Medicine, South West Healthcare, Warrnambool, VIC 3280, Australia
6 Deakin Medical School, Deakin University, Warrnambool, VIC 3280, Australia
Abstract
Anticoagulation using vitamin K antagonists or direct oral anticoagulants is an established treatment option for stroke prevention in patients with atrial fibrillation (AF). Although AF is common in patients with end-stage kidney disease (ESKD), the role of anticoagulation in stroke prevention has not been established in this group of patients. Major clinical trials have excluded patients with advanced kidney disease and this explains the significant lack of evidence-based guidelines to aid clinical decisions in the management of AF in patients with ESKD. Results from smaller studies and meta-analyses involving ESKD patients have not shown any significant advantage of using anticoagulants in preventing thromboembolic events. Moreover, anticoagulation has been associated with a higher risk of significant bleeding in dialysis patients. Therefore, caution and individualised treatment plans are suggested when considering ESKD patients’ anticoagulation. Ongoing clinical trials might illuminate this situation and help formulate more definitive guidance for anticoagulation use in ESKD patients. In summary, insufficient and inconclusive data, which results in the lack of evidence-based guidelines and the unique hemostatic paradox intrinsic to ESKD, muddle the management of AF in ESKD. This underscores the need to consolidate and synthesize data from past and ongoing studies to compare existing treatment options and identify gaps in knowledge that can direct further studies.
Keywords
- anticoagulation
- atrial fibrillation
- direct oral anticoagulants
- end-stage kidney disease
- stroke
- thromboembolism
- warfarin
End-stage kidney disease (ESKD) is a significant healthcare concern with increasing prevalence driven by factors such as ageing population, diabetes, heart disease and hypertension [1]. Patients with ESKD are particularly vulnerable to both thrombotic and haemorrhagic complications due to complex alterations in haemostatic pathways, and this presents a challenging paradox in clinical management [2]. These complications may manifest as serious events such as deep vein thrombosis, stroke and gastrointestinal haemorrhage, which significantly contribute to morbidity and mortality, highlighting the need for careful consideration in management [3].
Atrial fibrillation (AF) is another clinically significant comorbidity to consider in ESKD. The incidence of AF is reported to be two to three times higher in ESKD compared to the general population, affecting up to 20% of patients [4]. AF in this setting is associated with an increased risk of ischaemic stroke and systemic thromboembolism [5]. However, due to the unclear risk-benefit profile, a clear indication of anticoagulation in ESKD has not been established [6]. This ambiguity has led to variation in clinical practice among the treating physicians.
Unique differences in patients’ conditions make it hard to reliably weigh the
risk of thrombosis versus haemorrhage, leading to a dilemma in determining the
most appropriate approach to anticoagulation pharmacotherapy [3]. While existing scoring systems such as Congestive heart failure,
Hypertension, Age
This review aims to summarise the mechanisms of thrombotic and bleeding risk in ESKD, explore the challenges in anticoagulation management for AF in ESKD, and identify key directions for future research.
When characterising the predisposing events to thrombosis in ESKD, it is important to note that ESKD is often associated with comorbidities such as cardiovascular disease and diabetes mellitus, which mediate and/or accentuate the risk of thrombotic events [9]. The key predisposing features to thrombosis on a background of ESKD involve both the primary and secondary hemostatic processes and include changes such as endothelial damage, platelet activation and aggregation, increased coagulation factors and pro-thrombotic agents, and hypofibrinolysis [9] which are outlined in Fig. 1.
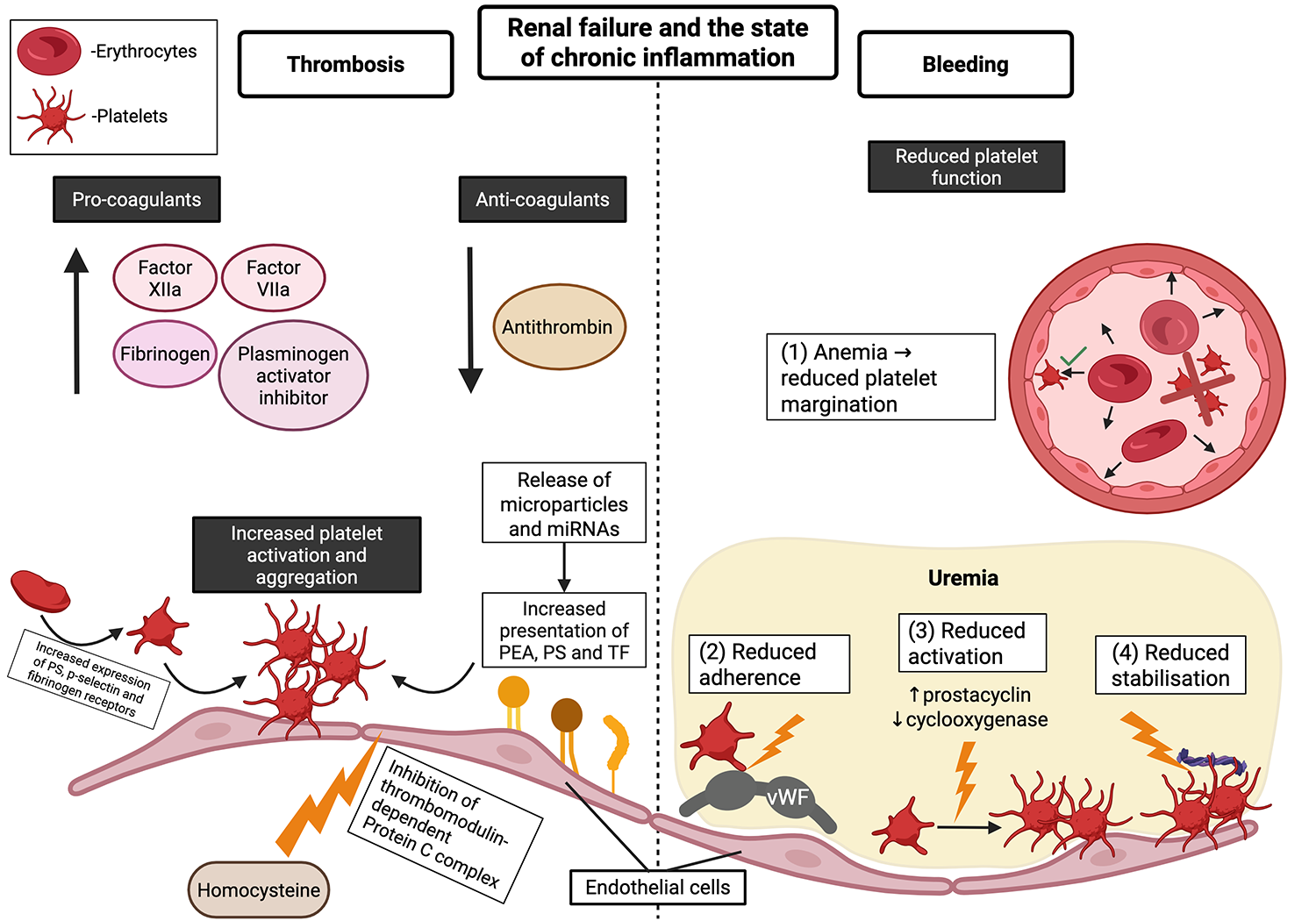 Fig. 1.
Fig. 1.
Pathophysiology of increased thrombosis and bleeding risk in end-stage kidney disease patients. This figure illustrates how renal failure and the state of chronic inflammation lead to both increased thrombosis (left panel) and increased bleeding (right panel), with platelets in various stages of activation (activated platelets have more membrane projections). Left panel: Increased levels of procoagulants, decreased levels of anticoagulants, and increased platelet activation and aggregation contribute to the increased risk of thrombosis. Right panel: Perturbations in multiple steps required for platelet function contribute to the increased risk of bleeding. The figure was created using BioRender (https://BioRender.comw00z483). PEA, phosphatidylethanolamine; PS, phosphatidylserine; TF, tissue factor; vWF, von Willebrand factor.
Renal failure is a chronic inflammatory state that induces endothelial cell damage and compromises its anti-thrombotic properties, thus setting the stage for downstream procoagulant events. Homocysteine has been reported to mediate the endothelial damage seen in individuals with renal dysfunction by inhibiting the thrombomodulin-dependent Protein C system [10]. This is followed by increased activation and aggregation of platelets [10] through increased expression of key receptors and proteins on the surface of platelets as highlighted in Fig. 1 [11]. In addition to the primary hemostasis process described above, the chronic inflammatory state in renal failure also modulates the secondary hemostasis process through modulating circulating levels of coagulation factors.
Previous studies have reported increased levels of coagulation factors such as fibrinogen, tissue factor (TF) [12], Factors XIIa, VIIa [13] and reduced levels of the anti-thrombotic agent, antithrombin [14]. Another system noted to play a contributory role is the renin-angiotensin-aldosterone system, as its activation is associated with raised levels of fibrinogen, D-dimer and plasminogen activator inhibitor (PAI) culminating in hypofibrinolysis [15]. In addition to these, there are other agents that have been identified to contribute to the prothrombotic state in renal failure.
Microparticles (MPs), formed from plasma membranes due to cell activation in a state of inflammation like that seen in renal disease, have been shown to promote the presentation of key proteins such as TF that kick off the coagulation cascade [16]. MPs have also been shown to play a role in regulating the function of platelets [17]. Other procoagulant agents include phosphatidylserine (PS) and phosphatidylethanolamine (PEA), which become exposed to the extracellular space during phospholipid bilayer scrambling [18].
ESKD patients are also more prone to a wide range of bleeding events, such as haemorrhagic stroke [19] and gastrointestinal bleeding [20]. This is due to pathophysiological mechanisms of chronic inflammation and increased oxidative stress, which impair multiple stages of the hemostatic process, leading to dysfunction in platelet recruitment, adhesion, activation, and aggregation as summarized in Fig. 1.
Platelet recruitment is affected by the anaemia prevalent in ESKD due to reduced erythropoietin production by the kidneys and inflammation, which causes chronic anaemia. Since red blood cells are required to transport platelets to sites of action, this decreases platelet recruitment [21]. In uremic patients, platelet adherence is impaired by reduced binding to von Willebrand factor (vWF) [10]. Uraemia also impairs platelet activation and aggregation by increasing endothelial prostacyclin [22] and decreasing cyclooxygenase [23]. Finally, platelet stabilisation is decreased by decreased platelet binding to fibrinogen [24].
Dialysis exacerbates this bleeding risk due to platelet activation and transient thrombocytopenia [25], which may explain one study’s finding that 34% of haemorrhagic strokes occurred in close temporal proximity to dialysis [26]. Aspirin and heparin are used to prevent vascular access thrombosis, and anticoagulation during dialysis further increases bleeding risk.
In summary, due to the overlapping mechanisms of bleeding in ESKD patients, anticoagulation therapy in this population must be administered with extreme caution.
The prevalence of AF is significantly higher in patients with ESKD compared to the general population [27]. It is estimated that AF occurs in approximately 1 out of 5 patients with ESKD, making it the most common cardiac arrhythmia in patients with advanced chronic kidney disease (CKD) [28, 29]. The underlying mechanisms connecting AF to CKD are thought to involve inflammatory markers and the renin-angiotensin-aldosterone system (RAAS) [30]. Inflammatory markers such as C-reactive protein (CRP) increase as CKD progresses, and increasing inflammation can lead to AF [31]. The activation of RAAS is a major factor in CKD progression as angiotensin II perpetuates glomerular capillary hypertension, which damages glomerular epithelial, endothelial, and mesangial cells [32]. Additionally, an angiotensin-converting enzyme (ACE) dependent increase in activated extracellular signal-regulated kinase 1/2 (Erk1 and Erk2) may contribute to atrial fibrosis, predisposing to the development of AF [33]. The traditional risk factors for AF, such as hypertension, heart failure, coronary artery disease, peripheral arterial occlusive disease and chronic obstructive pulmonary disorder, are also prevalent in ESKD [34].
Beyond these systemic mechanisms, several ESKD-specific factors also contribute to the incidence of AF in this population. Dialysis-related fluctuations in serum electrolytes, particularly potassium and calcium, can alter myocardial excitability and increase arrhythmia risk [35]. Furthermore, rapid intravascular volume shifts during dialysis could cause cardiovascular injury, while sympathetic nervous system activation triggered by ultrafiltration may further exacerbate atrial irritability, together increasing the risk of AF [36, 37].
A particularly concerning consequence of AF is the increased risk of thromboembolic events such as stroke, likely due to embolisation of thrombi formed in the left atrial appendage [38]. The annual absolute risk of stroke in patients with nonvalvular AF is 4%, and the presence of co-morbid conditions raises this risk by 15 to 20 times [39]. Aside from arterial thromboembolic events, a systematic review also reported significant short-term hazard ratios for venous thromboembolic events in AF at 6.18 and 9.62 for deep vein thrombosis and pulmonary embolism, respectively [40].
Anticoagulation use for stroke prevention is carefully weighed against its risk
of bleeding. Several scoring systems have been introduced to evaluate the risk
and indications for anticoagulation use for AF. The most common risk
stratification schemes are the CHA2DS2-VASc and HAS-BLED scores. The CHA2DS2-VASc
score is calculated from age, sex, and additional comorbidity data as follows: 2
points for history of prior stroke/transient ischemic attack or age
HAS-BLED is calculated by assigning 1 point for each of the following
components: hypertension (uncontrolled, systolic blood pressure
While these are useful in estimating bleeding risks in the general population, their applicability to patients with ESKD is limited due to suboptimal predictive performance. A key challenge is that the individual risk factors within these scores are weighted differently in patients of this population compared to the general population [7]. Using CHA2DS2-VASc may lead to inappropriate anticoagulation and increase bleeding risks, and HAS-BLED also lacks validity for predicting major bleeding for ESKD patients [7].
The Anticoagulation and Risk Factors in Atrial Fibrillation (ATRIA) bleeding
risk score offers another approach. It is calculated based on the presence of
anaemia (3 points), severe renal disease (3 points), age
These reflect the clinical challenge of objectively evaluating thromboembolic and bleeding risks in ESKD patients and underscore the need for further evaluation and validation of risk assessment tools for such patients.
Warfarin is a vitamin K epoxide reductase inhibitor that exerts its anticoagulant effects by inhibiting the synthesis of vitamin K-dependent coagulation factors. It has been used extensively for stroke prevention in patients with AF. Warfarin is primarily metabolised in the liver, and the kidneys eliminate a minimal amount. Moreover, warfarin is highly protein-bound and is not cleared during dialysis. Therefore, warfarin is pharmacokinetically suitable for patients with renal impairment, but concerns remain about its clinical effectiveness and safety in ESKD, with concerns about high bleeding risk. A 2016 systematic review and meta-analysis of 20 observational studies revealed that warfarin was not associated with any significant change in stroke outcomes but was associated with an increased risk of bleeding in patients with concomitant AF and ESKD [45].
Similarly, in another systematic review and meta-analysis involving dialysis patients, warfarin was not associated with any significant change in mortality or stroke. However, it was associated with an increased risk of major bleeding [46].
Randhawa et al. [47], in their systematic review and meta-analysis of 15 studies with 47,480 patients with AF and ESKD, concluded that warfarin use did not significantly reduce the risk of ischaemic stroke but was associated with a significantly higher risk of haemorrhagic stroke. There was no significant difference in the risk of other major bleeding and overall mortality [47].
A recent observational study involving 126 haemodialysis patients with AF showed that warfarin use was not associated with a lower risk of ischaemic stroke, haemorrhagic stroke or death in Indonesian patients [48].
Careful therapeutic dose monitoring may help decrease the bleeding risk, but this requires extensive follow-up and can be challenging to achieve. Even in strict trial conditions of RCTs, the average time in the therapeutic range hovers approximately 58% [49]. The use of warfarin in patients with ESKD or dialysis generally shows little mortality or thromboembolic benefits while increasing the risk of bleeding. However, this may also be due to the difficulty of maintaining warfarin in its therapeutic range.
Direct oral anticoagulants (DOACs) act by inhibiting coagulation factors such as thrombin (dabigatran) and factor Xa (edoxaban, rivaroxaban, apixaban) and are a popular alternative to warfarin due to their wider therapeutic window. Major clinical trials have shown that compared to warfarin, apixaban and rivaroxaban were superior or non-inferior to warfarin in preventing strokes in patients with AF with similar or better safety profiles. However, patients with stage 4 and 5 CKD were excluded from these trials [50, 51].
Unlike warfarin, all DOACs are eliminated by the kidneys in variable proportions. Nearly 80% of dabigatran elimination occurs through the kidneys, making it unsuitable for use in advanced renal impairment. Factor Xa inhibitors’ clearance is less dependent on renal excretion, accounting for 50% elimination for edoxaban, 35% for rivaroxaban, and 27% for apixaban. Rivaroxaban and apixaban are also highly protein-bound, 95% and 87%, respectively and are not removed during dialysis, making them more acceptable agents in ESKD patients [52].
Apixaban is usually preferred over other DOACs because it has the lowest renal clearance. However, data comparing DOACs to warfarin is sparse. Table 1 (Ref. [49, 53, 54, 55]) summarises the major RCTs, which are limited by the small sample size.
| Study name (Author, Year) | Study population | Study duration | Study arms (n) | Safety outcomes | Efficacy outcomes | TTR | Conclusion |
| Valkyrie [53] | Hemodialysis | February 2015–January 2019 | - Rivaroxaban 10 mg OD (46) - VKA INR 2–3 (44) - Rivaroxaban and vitamin K2 (42) |
- Bleeding: rivaroxaban HR 0.39 [0.17–0.90] | - Stroke/cardiac/vascular events: rivaroxaban HR 0.41 [0.25–0.68] | 48% | Rivaroxaban has better safety and efficacy than VKA |
| RENAL-AF [54] | Hemodialysis | January 2017–January 2019 | - Apixaban 2.5 mg BD/5 mg BD (82) - Warfarin INR 2–3 (72) |
- Bleeding (at 1 year): 32% in apixaban, 26% in warfarin (HR 1.20 [0.63–2.30]) | - Stroke/embolism (at 1 year): 3.0% [0.5%–9.7%] in apixaban, 3.3% [0.6%–10.5%] in warfarin | 44% (IQR 23%–59%) | Inadequate power due to recruitment challenges |
| AXADIA-AFNET 8 [55] | Hemodialysis | June 2017–May 2022 | - Apixaban 2.5 mg BD (48) - Phenprocoumon INR 2–3 (49) |
- Bleeding/death: 45.8% in apixaban, 51.0% in VKA (HR 0.93 [0.53–1.65]) | - Stroke/death/MI/thromboembolism: 20.8% in apixaban, 30.6% in VKA (p = 0.51) | 50.7% (range 0%–100%) | No apparent differences between apixaban and warfarin |
| SAFE-D [49] | Dialysis | December 2019–June 2022 | - Apixaban 5 mg BD (51) - Warfarin (52), No anticoagulation (48) |
- Bleeding: 2 in apixaban, 4 in warfarin, 2 in no anticoagulation | - 83% of participants completed follow-up in their allocated treatment arm | 58% (IQR 47%–70%) | Inadequate power (pilot study that proved feasibility of larger study) |
95% confidence intervals are indicated within square brackets.
Abbreviations: Valkyrie, Multicenter Randomized Controlled Trial of Vitamin K Antagonist Replacement by Rivaroxaban with or without Vitamin K2 in Hemodialysis Patients with Atrial Fibrillation; RENAL-AF, Renal Hemodialysis Patients Allocated Apixaban Versus Warfarin in Atrial Fibrillation; AXADIA-AFNET, Compare Apixaban and Vitamin K Antagonists in Patients With Atrial Fibrillation and End-Stage Kidney Disease; SAFE-D, Strategies for the Management of Atrial Fibrillation in Patients Receiving Dialysis; mg, milligrams; OD, once daily; VKA, vitamin K antagonist; INR, international normalised ratio; BD, twice a day; HR, hazard ratio; MI, myocardial infarction; TTR, Time in Therapeutic Range; IQR, interquartile range.
The Renal Hemodialysis Patients Allocated Apixaban Versus Warfarin in Atrial Fibrillation (RENAL-AF) trial was designed to compare apixaban 5 milligrams (mg) twice daily, reduced to 2.5 mg twice daily in patients above 80 years or weighing less than 60 kilograms (kg), versus warfarin in haemodialysis patients with AF. The trial was stopped prematurely, with a low sample size preventing investigators from drawing significant conclusions. This highlights the challenges of studying the effects of anticoagulation in this population. However, it is striking that the 1-year rates of bleeding events (approximately 30%) were 10 times higher than those of stroke or embolic events (3%) in both treatment arms [54].
A recently published Compare Apixaban and Vitamin K Antagonists in Patients with Atrial Fibrillation and End-Stage Kidney Disease (AXADIA-AFNET) 8 trial compared apixaban 2.5 mg twice daily with the vitamin K antagonist (VKA) phenprocoumon in 97 patients with AF and ESKD. There was no significant difference in the composite primary safety endpoints, defined as the first event of major bleeding, clinically relevant non-major bleeding, or all-cause mortality, or the primary efficacy outcome was ischemic stroke, all-cause death, myocardial infarction, and deep vein thrombosis or pulmonary embolism [55].
An extension of the Multicenter Randomized Controlled Trial of Vitamin K Antagonist Replacement by Rivaroxaban with or without Vitamin K2 in Hemodialysis Patients with Atrial Fibrillation (Valkyrie) trial involving 132 patients with AF on haemodialysis compared low-dose rivaroxaban (10 mg daily) with VKA (INR 2–3) or rivaroxaban (10 mg daily) and vitamin K2 (2000 µg menaquinone-7 thrice weekly after dialysis) and found that rivaroxaban had better safety and efficacy than VKAs, with a hazard ratio of 0.39 (95% confidence interval 0.17–0.90) and 0.41 (0.25–0.68) for bleeding and thromboembolism, respectively. Authors concluded that rivaroxaban, compared to VKAs, significantly decreased the composite outcome of fatal and nonfatal cardiovascular events and major bleeding complications in haemodialysis patients with AF [53].
A recent systematic review and meta-analysis of 4 randomised control trials, including 486 haemodialysis patients with AF, comparing DOACs with VKAs, showed no significant difference in ischemic stroke with relative risk (RR) 0.42; 95% confidence interval (CI) 0.17–1.04; p = 0.062; I2 = 0%, all-cause death (RR 0.88; 95% CI 0.57–1.35; p = 0.567; I2 = 47%), cardiovascular death (RR 1.13; 95% CI 0.60–2.10; p = 0.700; I2 = 0%), or clinically relevant non-major bleeding (RR 1.11; 95% CI 0.67–1.84; p = 0.669; I2 = 0%). However, a statistically significant reduction in major bleeding was noted in the DOAC group (RR 0.64; 95% CI 0.41–0.98; p = 0.044; I2 = 0%) [56].
Another recent meta-analysis of 42 studies, including 3 randomised trials involving 185,864 ESKD patients with AF comparing DOACs, VKAs, and no anticoagulation, showed that VKAs were associated with a significantly higher risk of major bleeding compared to DOACs, with hazard ratio (HR) 0.74 (95% CI 0.64–0.84) or no anticoagulation (HR 1.47; 95% CI 1.34–1.61). Of note, there was no difference in preventing thromboembolism between VKAs, DOACs, and no anticoagulation [57].
There is no consensus among international guidelines regarding the use of DOACs in ESKD patients. Most guidelines recommend making decisions on a patient-to-patient basis, weighing the pros and cons of anticoagulation in individual patients. The Kidney Disease Improving Global Outcomes (KDIGO) work group suggests apixaban at a reduced (2.5 mg twice daily) dose in this group of patients [58].
National Institute for Health and Care Excellence (NICE) guidelines state that aspirin should not be offered as monotherapy for thromboembolism prevention in AF due to its limited efficacy in stroke prevention and increased risk of bleeding [59]. In ESKD and dialysis, the use of single antiplatelet therapy with aspirin is associated with variable bleeding risk [60]. Combination treatment with aspirin and warfarin significantly increases the risk of bleeding. Therefore, caution should be exercised in patients with ESKD and AF [61].
Dual antiplatelet therapy (DAPT) with aspirin and clopidogrel may be considered for patients with AF who are unsuitable for warfarin therapy. However, it is shown to have inferior stroke prevention compared to warfarin in the Atrial fibrillation Clopidogrel Trial with Irbesartan for prevention of Vascular Events (ACTIVE W) study [62]. Furthermore, dual antiplatelet therapy has also been shown to have an increased risk of significant bleeding in patients with kidney disease on haemodialysis [63].
Left atrial appendage occlusion (LAAO) is a minimally invasive transcatheter procedure in which a device is implanted to seal off the left atrial appendage to prevent clot formation [64]. Although further research is needed to determine its safety and efficacy, studies suggest that LAAO may be an effective alternative for stroke prevention in AF in the ESKD population [65, 66].
Pharmacodynamics and pharmacokinetics research have played key roles in developing existing therapies and testing their effects in the absence of available clinical data. They could represent the next step in developing and testing alternative therapies that are less affected by renal clearance, have a wider therapeutic window, or have more predictable effects that are amenable to easy titration.
Meanwhile, more RCTs are needed to guide decision-making on whether and how to anticoagulate patients with concomitant AF and ESKD. Specifically, scoring systems such as CHA2DS2-VASc, HAS-BLED, and ATRIA require validation in this especially vulnerable population. However, this presents significant challenges, and an alternative is to standardise the study design of non-randomised prospective and retrospective observational studies to become more amenable to meta-analysis.
Upcoming RCTs are summarised in Table 2. The Danish Warfarin-Dialysis Study-Safety and Efficacy of Warfarin in Patients With Atrial Fibrillation on Dialysis (DANWARD) is projected to be completed in 2027 (NCT03862859) [67]. Quite uniquely, Safety and Efficacy of Apixaban Versus Warfarin in Peritoneal Dialysis Patients with Non Valvular Atrial Fibrillation: a Prospective, Randomised, Open-label, Blinded End-point Trial (APIDP2), an RCT investigating patients on peritoneal dialysis and their response to warfarin and apixaban, is projected to be completed in 2027 (NCT06045858) [68]. Stroke Prophylaxis With Apixaban in Chronic Kidney Disease Stage 5 Patients With Atrial Fibrillation (SACK) is projected to be completed in 2028 and includes patients with and without dialysis (NCT05679024) [69].
| Study name (NCT) | Projected completion | Study population | Study duration | Study arms | Study total (n) | Outcomes |
| APIDP2 (NCT06045858) | 2027 | Peritoneal dialysis | October 2024–October 2027 | - Apixaban 2.5 mg BD | 178 | - Safety: major or clinically relevant non-major bleeding |
| - Warfarin INR 2–3 | - Efficacy: stroke or TIA, cardiovascular death and thrombosis including myocardial infarction | |||||
| DANWARD (NCT03862859) | 2027 | Dialysis | October 2019–January 2026 | - Warfarin INR 2–3 | 718 | - Safety: major bleeding |
| - No anticoagulation | - Efficacy: stroke or TIA | |||||
| SACK (NCT05679024) | 2028 | CKD stage 5 with or without dialysis | February 2023–December 2028 | - Apixaban 2.5 mg BD | 1000–1400 | - Safety: fatal, intracranial, or major bleeding |
| - No anticoagulation | - Efficacy: ischemic stroke, cardiovascular event, thromboembolism, mortality |
Abbreviations: NCT, National Clinical Trial; APIDP2, Safety and Efficacy of Apixaban Versus Warfarin in Peritoneal Dialysis Patients With Non Valvular Atrial Fibrillation: a Prospective, Randomised, Open-label, Blinded End-point Trial; DANWARD, The Danish Warfarin-Dialysis Study-Safety and Efficacy of Warfarin in Patients With Atrial Fibrillation on Dialysis; SACK, Stroke Prophylaxis With Apixaban in Chronic Kidney Disease Stage 5 Patients With Atrial Fibrillation; CKD, chronic kidney disease; mg, milligrams; BD, twice a day; INR, international normalised ratio; TIA, transient ischaemic attack.
The Strategies for the Management of Atrial Fibrillation in Patients Receiving Dialysis (SAFE-D) trial, a recently concluded pilot study investigating warfarin and apixaban in dialysis patients with AF, showed that it is feasible to recruit and measure outcomes in this patient population [49]. Therefore, future RCTs may shed some light on using warfarin and apixaban in ESKD. However, there is still a paucity of data on the other DOACs and alternatives such as aspirin.
In conclusion, anticoagulation agents’ safety and efficacy profiles must be further characterised in ESKD patients to equip clinicians with better AF management strategies. The greater prevalence of AF and higher risk of complications associated with AF in patients with ESKD and the exclusion of this population from clinical trials, as well as scoring systems, underscore the need to establish AF management guidelines specific to those with ESKD. While there is a considerable spotlight on the use of DOACs for the management of AF, their wider therapeutic window and their association with reduced embolic phenomena, its major criticism seems to be that they do not exhibit similar superiority in reducing major bleeding events, especially gastrointestinal bleeding. Considering that DOACs have substantial renal clearance, the pathophysiology of the haematological paradox in ESKD further muddles the picture and the ability to gauge the relative effectiveness of DOACs. The acknowledgement that factors such as ethnicity and age also modify an individual’s predisposition to thrombosis and/or bleeding highlights the importance of personalising AF management strategies beyond simply an individual’s medical history. Hopefully, the upcoming RCTs mentioned above may shed light on the use of warfarin, DOACs, and other alternatives, such as aspirin, to manage AF in patients with ESKD.
Not applicable.
SSYW, DP, KM, XRL, RGI, and MMJ equally contributed in literature search, data gathering, data interpretation, and drafting the manuscript. SSYW and MMJ designed the review. XRL created the figure for the article. All authors contributed to revising the manuscript critically for important intellectual content. All authors read and approved the final manuscript. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
This research received no external funding.
Fig. 1 was created using BioRender. The authors have no financial or personal relationship with BioRender, and the use of this tool does not imply any endorsement. The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

