1 Department of Gastroenterology, Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, 430030 Wuhan, Hubei, China
Abstract
Autoimmune pancreatitis (AIP) is a relatively rare chronic fibroinflammatory disorder of the pancreas caused by autoimmune mechanisms. Patients with this condition generally show a clear response to glucocorticoid therapy. Notably, its clinical and imaging features often resemble those of pancreatic cancer (PC), particularly when AIP presents as a focal mass. In such cases, clinicians may confuse the two diseases. Given that AIP and PC differ considerably with respect to their biological behavior and treatment, diagnostic errors can lead to unnecessary surgery or delayed treatment. Consequently, it is essential to accurately distinguish between these conditions. Recent diagnostic advances, including the application of liquid biopsy and artificial intelligence, are now being evaluated as alternative approaches to conventional diagnostic methods, and may contribute to improving the distinction between AIP and PC. In this review, we summarize the current evidence, outline the clinical profile of AIP, and compare AIP with PC with respect to epidemiological, clinical, serological, imaging, and histopathological dimensions. In addition, we discuss the advantages and limitations of these new diagnostic tools. Furthermore, we propose a practical three-stage diagnostic algorithm based on the present guidelines. This stepwise approach may provide a practical method for integrating routine and emerging tests for evaluating patients with suspected AIP or PC.
Keywords
- autoimmune pancreatitis
- pancreatic cancer
- differential diagnosis
- imaging
- liquid biopsy
- artificial intelligence
Autoimmune pancreatitis (AIP) is a chronic fibroinflammatory disease of the pancreas caused by autoimmune responses, which is highly responsive to corticosteroid therapy. Although Sarles et al. [1] first described this condition in 1961, it was not until 1995 that the concept of “autoimmune pancreatitis” was explicitly proposed and widely recognized [2]. Clinically, AIP has no specific signs or symptoms, and a majority of the initial symptoms, including painless obstructive jaundice, abdominal pain, and weight loss, are similar to the clinical manifestations of pancreatic cancer (PC). Radiologically, AIP often manifests as a diffuse or focal pancreatic enlargement with irregular or segmental ductal narrowing. Such radiological and clinical overlap, particularly with respect to focal lesions, makes it difficult to distinguish between these two diseases.
Previous studies have highlighted this diagnostic challenge. For example, the International Study Group of Pancreatic Surgery has reported that 5% to 13% of patients undergoing pancreatic resection for suspected malignancy were ultimately found to have benign disease, with AIP accounting for approximately 30% to 43% of these cases [3]. In addition, a recent meta-analysis of 8917 patients who underwent pancreatic resection for suspected cancer revealed that 10.1% of the specimens were histologically benign, with AIP constituting approximately 26% of all unnecessary surgeries [4]. Differentiating AIP from pancreatic ductal adenocarcinoma (PDAC) remains challenging even when applying modern diagnostic methods. However, given the fundamental differences in the nature of AIP and PC and the respective treatment strategies, accurate identification is essential to avoid misdiagnosis.
With the advent of artificial intelligence (AI), radiomics, liquid biopsy, and multi-omics integration, diagnostic approaches have shifted from morphology-based assessments to comprehensive, multidimensional, investigational strategies. In this review, we systematically compare AIP and PC with respect to clinical, serological, imaging, and histopathological domains, and critically evaluate the roles played by emerging diagnostic tools and the challenges associated with their application. Moreover, building upon the current guidelines, we propose a practical three-stage diagnostic algorithm. This framework clearly outlines the integration of serological tests, imaging, endoscopic ultrasound (EUS)-guided tissue sampling, steroid trials, and novel technologies into a single clinical workflow. Our goal in presenting this review is to provide clinicians with a clear and actionable strategy that may help improve the differentiation between these two diseases.
Here, we present a narrative review focusing on the differentiation between AIP and PC. A systematic search was conducted in PubMed (https://pubmed.ncbi.nlm.nih.gov/) and Web of Science (https://www.webofscience.com/) to identify relevant English-language articles published between 1 January 2020 and 31 October 2025. Our search strategy combined keywords and Medical Subject Headings (MeSH) related to the target conditions (“autoimmune pancreatitis”, “AIP”, “IgG4-related disease”, “IgG4-RD”, “pancreatic cancer”, “pancreatic ductal adenocarcinoma”) with terms covering diagnostic modalities and emerging technologies, including “CT”, “MRI”, “MRCP”, “EUS”, “biopsy”, “steroid trial”, “liquid biopsy”, “radiomics”, “artificial intelligence”, “machine learning”, and “deep learning”. To ensure the comprehensiveness of the literature search, we manually screened the reference lists of key articles and clinical guidelines. This step allowed us to include seminal articles published before 2020, when relevant to provide the necessary background context.
At present, the International Consensus Diagnostic Criteria (ICDC), proposed in 2011, remains the most widely applied framework for diagnosing AIP. The ICDC incorporate five diagnostic aspects, namely, pancreatic imaging, serum immunoglobulin G4 (IgG4) status, involvement of other organs, histopathology, and steroid responsiveness, on the basis of which AIP is categorized into two major forms, type 1 and type 2 [5]. It is worth noting that since the establishment of the ICDC standards, a number of international authoritative societies have successively issued updated guidelines and consensus statements, which have contributed to providing a more in-depth review and specification of AIP and similar immunoglobulin G4-related disease (IgG4-RD). These include the IgG4-RD classification criteria developed by the European Federation of Gastroenterology, Swiss Society of Gastroenterology [6], Japanese Pancreas Society [7], the American College of Rheumatology, and the European League Against Rheumatism in 2019 [8]. Collectively, these documents have led to the standardized diagnosis and management of this disease spectrum.
Type 1 AIP, also referred to as lymphoplasmacytic sclerosing pancreatitis, occurs predominantly in older men and is closely associated with IgG4-RD [5], and is commonly characterized by elevated levels of serum IgG4 and multi-organ involvement, including biliary, salivary gland, and renal manifestations [6, 8]. Type 2 AIP, also referred to as idiopathic duct-centric pancreatitis, more often affects younger individuals, lacks gender predominance, and is typically confined to the pancreas. In addition, approximately 30% of the patients with type 2 AIP also present with concomitant inflammatory bowel disease (IBD) [5], with a prospective study focusing on type 2 AIP reporting a high co-occurrence with IBD, the incidence of which during the long-term follow-up was 87.5% [9]. However, in patients with type 2 AIP, serum IgG4 levels generally remain within the normal range [5].
Imaging of both subtypes commonly reveals diffuse or localized pancreatic enlargement, with a classic “sausage-shaped” morphology, capsule-like rim, and delayed contrast enhancement [7]. Moreover, both these subtypes are highly responsive to corticosteroid therapy, with excellent rates of remission, although relapse is common. A large international multi-center retrospective study has reported remission rates of 99% for type 1 AIP and 92% for type 2 following steroid therapy, with a significantly higher rate of type 1 recurrence compared with that of type 2 (31% vs. 9%) [10]. Consistently, a meta-analysis of 36 studies has reported that patients with type 1 AIP had higher rates of recurrence than those with type 2 (37.5% vs. 15.9%) [11]. The key distinguishing clinical, serological, imaging, and histological features of type 1 AIP, type 2 AIP, and PC are summarized in Table 1 (Ref. [5, 6, 7, 8, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21]).
| Feature | Type 1 AIP | Type 2 AIP | Pancreatic cancer |
| Typical onset age | Around 64.8 years, older males [12] | Around 43 years, no gender bias [13] | |
| Serum IgG4 | Elevated in most cases (87.8%) [5, 16] | Normal or rarely elevated [7, 13] | Elevated in 10% of cases [17] |
| Extra-pancreatic involvement | Common (biliary, salivary glands, kidneys) [6, 8] | Rare [5] | Due to metastasis (liver, lungs, lymph nodes) [18] |
| Histology | Lymphoplasmacytic infiltration, storiform fibrosis, obliterative phlebitis [5] | Granulocytic epithelial lesions, neutrophil infiltration [5] | Atypical glands, desmoplastic stroma, vascular invasion [19] |
| Associated diseases | Often IgG4-RD [6, 8] | Associated with IBD (about 30%) [5] | - |
| Imaging features | Diffuse/focal enlargement, capsule-like rim, delayed enhancement [7, 20] | Similar to type 1 AIP [7, 13] | Irregular mass, ductal obstruction, parenchymal atrophy [20, 21] |
| Response to steroids | Excellent (99% remission rate) [10] | Excellent (92% remission rate) [10] | No response |
| Relapse rate | High (about 31%–38%) [10, 11] | Low (about 9%–15.9%) [10, 11] | Not applicable |
Note: The proposed “type 3 AIP” is not included as it is not yet a formally recognized entity in consensus classifications. Abbreviations: AIP, autoimmune pancreatitis; IgG4, immunoglobulin G4; IgG4-RD, immunoglobulin G4-related disease; IBD, inflammatory bowel disease.
AIP not otherwise specified (AIP-NOS) refers to a subtype of AIP that does not meet the criteria for type 1 or type 2 [5]. Patients with this form typically lack histological confirmation and have elevated levels of serum IgG4, extra-pancreatic involvement, or associated IBD, and although not recognized as a distinct pathological entity, AIP-NOS accounts for approximately 16% of AIP cases [22].
More recently, in 2023, Thomas et al. [23] introduced the concept of type 3 AIP, which was proposed as an immune checkpoint inhibitor–associated form of pancreatitis. This variant is induced by non-specific inflammatory T-cell responses targeting the pancreatic ducts and acini, most often in patients with advanced malignancies, such as melanoma and lung cancer [23]. These patients are frequently asymptomatic or mildly symptomatic, and generally have normal serum IgG4 levels, although occasionally, these may be transiently elevated. Histologically, type 3 AIP is characterized by a predominantly cluster of differentiation 8 (CD8)-positive T-cell infiltration without distinct diagnostic features [23]. However, it should be noted that the “type 3 AIP” classification remains a descriptive research concept and has not been formally incorporated into the ICDC.
Whereas type 1 AIP and PC occur predominantly in older men, type 2 typically presents earlier, with a mean age of onset of approximately 43 years, and shows no obvious gender predominance [13]. Type 1 AIP is considerably more common than type 2, accounting for 96% of the cases in Japan and approximately 80% in Western countries [24]. Epidemiologically, a 2016 nationwide survey in Japan reported that the incidence of type 1 AIP is 1 to 3 per 100,000 adults, with a prevalence of approximately 10 per 100,000, a male-to-female ratio of 2.94:1, and a mean age at diagnosis of 64.8 years [12]. Comparatively, PC tends to be more common in Western countries, and the risk of developing PC increases markedly with age. According to the Global Cancer Observatory 2020 estimates [14], PC annually accounts for approximately 500,000 new cases and 470,000 deaths globally, ranking seventh in incidence, although third to fourth in terms of cancer-related mortality, with a dismal overall 5-year survival rate of only approximately 10% to 12%. Furthermore, the rates of both incidence and mortality are higher in males than in females. In the United States, the average age at diagnosis for PC is 68 years, as reported by the National Cancer Institute’s Surveillance, Epidemiology, and End Results program of the U.S. National Cancer Institute [15]. In addition, data obtained in China in 2019 indicated that age-standardized disability-adjusted life years for PC peaked in the 70–74 age group [25].
Both AIP and PC lack specific clinical manifestations, with obstructive jaundice being the most common initial manifestation [12]. Patients with AIP and PC may, however, show subtle differences with respect to the manifestations of jaundice, with that caused by AIP fluctuating or even spontaneously regressing, whereas the jaundice caused by PC is generally progressive [7]. Moreover, the two diseases tend to be characterized by distinctly different patterns of extra-pancreatic involvement, which in type 1 AIP frequently involves the biliary tract, salivary glands, and other organs as part of IgG4-RD [26]. In contrast, distant metastases of PC occur primarily in the liver, lungs, peritoneum, omentum, and lymph nodes [18].
Although serum IgG4 generally serves as a key biomarker in the diagnosis of type
1 AIP, its sensitivity is relatively low. Approximately 10% of patients with PC
have elevated levels of this marker [17], and the findings of a recent
meta-analysis of 13 studies revealed that serum IgG4 had a sensitivity of 72%
and specificity of 93% for distinguishing AIP from PC [27]. Furthermore, a
national survey conducted in Japan in 2024, which included only patients with
type 1 AIP, revealed that 87.8% of these patients were serum IgG4 positive
(
Although carbohydrate antigen 19-9 (CA19-9) is the most widely used tumor marker for PC, it remains undetectable in approximately 10% of individuals who lack the Lewis antigen and may show a slight increase in patients with chronic pancreatitis [28]. Notably, a recent study indicated that Lewis-negative PC patients have a poorer prognosis and higher risk of metastasis [29], and a meta-analysis has reported pooled sensitivity and specificity values of 74.8% and 78.2%, respectively, for CA19-9 with respect to the diagnosis of PDAC [30].
Given the limitations of individual markers, combining biomarkers has shown promise regarding improvements in differential diagnosis. For example, van Heerde et al. [31] have demonstrated that a combination of IgG4 and CA19-9 can have sensitivities and specificities of up to 94% and 100%, respectively, in distinguishing AIP from PC, notably outperforming either marker applied alone. Furthermore, a recent single-center study reported that the CA19-9/direct bilirubin ratio can be used to differentiate CA19-9-elevated pancreatic head AIP from pancreatic head cancer, with an area under the curve (AUC) of 0.772 and sensitivities and specificities greater than 70% [32]. However, these findings were based on retrospective, highly selected cohorts and, accordingly, require prospective validation in larger, more diverse populations.
In addition to different autoantibodies, serum levels of IgG2, soluble interleukin-2 receptor, and C-C motif chemokine ligand 18 also tend to be elevated in patients with AIP compared to healthy individuals [33], and it has been suggested that these can be used as novel biomarkers of IgG4-RD; however, large-scale studies are needed by way of confirmation.
Imaging serves as the first-line method for distinguishing AIP from PC. AIP may present with focal, multifocal, or diffuse pancreatic involvement, with the focal form presenting the greatest diagnostic difficulty [5]. The most common site of a focal mass of AIP is the head of the pancreas, followed by the body and the tail [9]. Similarly, a study has found that approximately 75% of PC lesions are located in the head of the pancreas, 17% to 26% in the body/tail of the pancreas, and rarely involve the entire pancreas [18]. Notably, however, advances in imaging techniques have provided new perspectives and methods for differential diagnosis.
Computed tomography (CT) and magnetic resonance imaging (MRI) are the primary imaging modalities for evaluating pancreatic parenchymal disease. Classic features of AIP include diffuse pancreatic enlargement, a “sausage-like” appearance, capsule-like rim, and delayed homogeneous enhancement [7], whereas, comparatively, PC generally appears as a localized mass characterized by ill-defined borders and irregular or minimal contrast uptake [34]. A meta-analysis conducted by Yoon et al. [35], who comprehensively synthesized the CT features of patients with AIP and PC revealed that diffuse enlargement, delayed homogeneous enhancement, a capsule-like rim, multiple pancreatic lesions, renal involvement, retroperitoneal fibrosis, and biliary abnormalities favored a diagnosis of AIP, whereas discrete masses, abrupt ductal cutoff, upstream ductal dilatation, and parenchymal atrophy were more suggestive of PC. Similarly, a further meta-analysis conducted by the same group focusing on MRI features revealed that diffuse enlargement, a capsule-like rim, multiple ductal strictures, and uniformly delayed enhancement were more indicative of AIP, whereas discrete masses and ring-like enhancement were more typical of PC [20]. In addition, Lee et al. [36] further demonstrated that MRI provides better discriminatory performance than CT in differentiating focal AIP from PC.
Diffusion-weighted imaging (DWI) is a widely adopted technique used to assess pancreatic lesions, which acquires images at different b-values and facilitates calculation of the apparent diffusion coefficient (ADC) that provides a quantitative measure of water diffusion. In general, a higher DWI signal corresponds to a lower ADC. In this regard, Ren et al. [37] have reported that the average ADC value in mass-forming AIP is markedly lower than that in PC, reflecting the restricted water diffusion in the former, and the findings of a meta-analysis have indicated that DWI can achieve a pooled sensitivity of 82% and specificity of 84% for differentiating AIP from PC [38]. However, a standardized ADC value cutoff for AIP has yet to be established [37, 39]. It is probable that differences in this regard arise from differences in DWI acquisition parameters, particularly with respect to b-value selection and disease activity. Further work is accordingly needed to standardize DWI protocols. In addition, magnetic resonance elastography, which assesses tissue stiffness, has shown promise in enhancing diagnostic differentiation [40].
Endoscopic retrograde cholangiopancreatography (ERCP) and magnetic resonance
cholangiopancreatography (MRCP) are primarily used to evaluate the
pancreatobiliary ductal system and provide high sensitivity for detecting ductal
abnormalities. However, given its non-invasiveness, MRCP has largely supplanted
diagnostic ERCP [41]. The characteristic ductal features of AIP include long
segment (
EUS provides clear images of the parenchymal and ductal structures, and has excellent sensitivity and specificity for pancreatic diseases [44]. Most importantly, fine-needle aspiration or biopsy (FNA/FNB) can be performed simultaneously with EUS to obtain histological specimens. In AIP, EUS typically reveals diffuse hypoechoic enlargement, duct-penetrating signs, irregular ductal narrowing, and biliary wall thickening. The classic finding is a “sausage-like” pancreas with diffuse hypoechoic enlargement and heterogeneous internal echogenicity [45, 46]. In contrast, PC generally presents as a localized lobulated hypoechoic mass, frequently associated with the double-duct sign, regional lymphadenopathy, or vascular invasion [47].
In recent years, the development of several advanced EUS-based techniques has contributed to enhancing diagnostic capabilities. Contrast-enhanced ultrasound is used to evaluate blood flow in pancreatic masses [48], whereas endoscopic ultrasound elastography assesses the stiffness and elasticity of pancreatic lesions [46], and contrast-enhanced harmonic EUS can be used to provide detailed visualization of the microvascular architecture and blood flow within tissues [49, 50]. All of the aforementioned techniques can be used for the diagnosis and differential diagnosis of pancreatic diseases and are considered to have promising clinical potential. However, studies that have assessed these techniques have generally been based on small numbers of cases and non-randomized controlled designs, and thus, further research is needed to confirm their applicability for differential diagnosis.
Positron emission tomography-computed tomography (PET-CT) is an imaging approach that facilitates simultaneous assessment of both the pancreas and other organs. Both AIP and PC are characterized by heightened 18F-fluorodeoxyglucose (18F-FDG) uptake on PET-CT, with the former typically showing diffuse or multifocal uptake, whereas the latter generally presents with focal nodular uptake [51]. Moreover, the maximum standardized uptake value tends to be higher in PC than in AIP [52].
In addition to FDG, researchers in the field of molecular imaging are actively assessing the utility of more specific tracers. For example, given that fibrosis serves as a hallmark of IgG4-RD, 68Ga-fibroblast activation protein inhibitor (FAPI) PET, which targets fibroblast activation proteins, has been evaluated for its application in the diagnosis of IgG4-RD. A single-center prospective study, in which the authors compared 68Ga-FAPI PET-CT with FDG PET-CT to detect organ involvement in IgG4-RD, revealed that compared with 18F-FDG, there was significantly higher uptake of 68Ga-FAPI in the pancreas, bile duct/liver, and salivary glands [53]. In addition, the findings of a further study revealed that compared with FDG PET-CT (72.5%), 68Ga-prostate-specific membrane antigen (PSMA) PET-CT achieved greater accuracy in distinguishing PC from AIP (92.5%) [54], thereby highlighting the potential of these targeted probes. However, these findings were based on assessments using small single-center cohorts. Accordingly, current estimates of the clinical efficacy of 68Ga-PSMA and 68Ga-FAPI PET-CT remain preliminary and require validation in larger multi-center populations before these techniques can be incorporated into routine diagnostic strategies.
Although histopathology remains the gold standard for distinguishing AIP from PC, the invasive nature and sampling limitations restrict its use in routine clinical practice. As more viable alternatives, EUS-guided tissue acquisition has emerged as the principal method for obtaining pathological specimens, and whereas FNA is typically employed for obtaining cytological specimens with limited tissue, FNB can facilitate the collection of core tissues that preserve histological structures, thereby enabling the identification of characteristic features such as lymphoplasmacytic infiltration and obliterative phlebitis [55]. In this context, a meta-analysis confirmed that EUS-FNB provides significantly higher diagnostic accuracy (87.2%) than FNA (55.8%) [56]. However, despite recent advances, EUS-guided AIP sampling continues to present certain challenges. For example, the dense fibrosis and patchy inflammation observed in AIP often make it difficult to obtain adequate tissue cores, even when using FNB needles [57, 58]. The quality of the samples obtained also tends to be operator dependent. Moreover, factors such as the number of needle passes, use of suction or slow-pull techniques, and access to on-site cytopathology can influence the diagnostic yield.
Comparatively, EUS diagnosis of PDAC typically reaches an accuracy of
Recent findings have, nevertheless, been encouraging. For example, a multi-center prospective study that assessed the utility of end-cutting needles for EUS-FNB in 52 patients with suspected AIP reported an improved diagnostic yield in 92.3% of the cases according to ICDC criteria. A definitive diagnosis was made for 74.3% of the patients and a probable diagnosis for 17.9%. This technique has also been established to distinguish type 1 AIP from type 2 AIP, exclude malignancy with 100% specificity, and achieve an overall diagnostic accuracy of 94.2% [60].
The classic histological hallmarks of type 1 AIP include dense lymphoplasmacytic infiltration, storiform fibrosis, obliterative phlebitis, and more than 10 IgG4-positive plasma cells per high-power field [7]. In contrast, type 2 AIP is characterized by granulocytic epithelial lesions and prominent neutrophilic infiltration [13].
In cases in which histology is unclear but EUS-guided biopsy reasonably excludes malignancy, a short diagnostic trial of corticosteroids can be considered. A standard regimen uses oral prednisone at 0.6–1 mg/kg per day for 2 weeks, and numerous AIP patients who have received this treatment have been found to show clear clinical and radiological improvements [5]. However, for cases in which PC is highly suspected, clinicians should refrain from initiating steroid trials, as inappropriate glucocorticoid use in patients with undiagnosed PDAC can mask symptoms and inflammatory markers, which can delay diagnosis, curative surgery, or systemic therapy. Experimental evidence further indicates that in PDAC models, glucocorticoids may contribute to chemoresistance and tumor progression [61, 62, 63]. Consequently, current guidelines recommend short-term steroid trials for carefully selected patients, and only after the rigorous exclusion of malignancies and under close multidisciplinary supervision. In this setting, although a positive steroid response supports the diagnosis of an AIP, this ideally needs to be evaluated in conjunction with histological, imaging, and serological findings rather than being used independently.
Liquid biopsy is increasingly being recognized as a valuable and less invasive tool for identifying and monitoring pancreatic diseases. By analyzing circulating tumor cells (CTCs), circulating cell-free DNA (cfDNA), circulating tumor DNA (ctDNA), extracellular vesicles, such as exosomes, and microRNAs (miRNAs) in the blood or other bodily fluids, this technique facilitates the characterization of tumor biology without invasive tissue acquisition [64]. Compared with conventional tissue biopsies, liquid biopsy has unique advantages in capturing tumor heterogeneity and detecting early-stage lesions [65, 66].
A range of liquid biopsy markers has been shown to have promising diagnostic potential. For example, a prospective study of a cohort of 80 patients revealed that combining CTCs with CA19-9 achieved an area under the curve (AUC) of 0.95, which was markedly superior to that achieved using CA19-9 alone (AUC = 0.80) [67]. In addition, ctDNA, the tumor-specific fraction of cfDNA, has been extensively studied as a promising biomarker of PC. Nishizawa et al. [68], for example, have reported that hypermethylation of the cysteine dioxygenase type 1 promoter region in PDAC tissues can be used to distinguish tumors from adjacent non-neoplastic samples, with an AUC of 0.97. Similarly, Berger et al. [69] measured ctDNA and combined CA19-9 and thrombospondin-2 protein levels to successfully discriminate among PDAC, intraductal papillary mucinous neoplasms, and chronic pancreatitis, achieving an AUC of 0.94. Furthermore, exosomes, which are highly stable, have demonstrated a sensitivity of 79%, specificity of 77%, and an AUC of 0.85 in differentiating pancreatic cancer from non-cancer controls using miRNA signatures [70].
Multi-marker panels are increasingly being demonstrated to outperform single biomarkers, and the integration of genomic, transcriptomic, and proteomic signatures with machine learning (ML) algorithms has facilitated superior diagnostic performance. For example, Ben-Ami et al. [71] have demonstrated that a combined assay of protein biomarkers and differentially methylated cfDNA can achieve a sensitivity exceeding 90% for the early detection of PC, which was considerably higher than the level obtained using CA19-9 alone. Similarly, Zhao et al. [72] have reported that the application of cfDNA methylation integrated with other molecular data achieved a sensitivity of 86% and specificity of 92% for the early detection of PDAC. Furthermore, Cohen et al. [73] developed a multi-analyte blood test (CancerSEEK) combining ctDNA mutations with circulating protein markers, achieving specificities exceeding 99% and sensitivities ranging from 69% to 98% for multiple surgically resectable cancers, including PC.
Recent evidence has further validated the utility of liquid biopsies. A comprehensive meta-analysis of 1872 cases revealed excellent overall diagnostic performance, in which this technique was used to achieve 80% sensitivity, 89% specificity, and an AUC of 0.93, with exosome-based evaluations performing even better, reaching 93% sensitivity, 92% specificity, and an AUC of 0.98 [74]. In addition, the findings of a Circulating Cell-free Genome Atlas study confirmed that cfDNA methylation testing can achieve specificities in excess of 99%, with 43.9% sensitivity for stage I–III cancers and 93% accuracy in tumor tracing [75].
Other novel biomarkers are also being investigated, as demonstrated by Martínez-Bosch et al. [76], who showed that levels of soluble AXL receptor tyrosine kinase protein in serum had a high specificity for distinguishing PC from chronic pancreatitis, whereas Guo et al. [77] have identified unique lipid metabolic signatures in the exosomes of patients with PC.
Although less invasive than other techniques, the clinical application of liquid biopsy for differentiating AIP from PC faces multiple challenges, among which a key problem is a lack of standardization. Differences in sample type, processing, and storage can have a significant influence on sample quality [78, 79, 80], whereas the diversity of analytical methods, such as quantitative polymerase chain reaction (qPCR), digital PCR (dPCR), and next-generation sequencing, and their differing performance characteristics make comparison difficult [81, 82]. Moreover, a lack of unified cutoff values for biomarkers, limited quality control among laboratories, and unclear reimbursement policies limit their clinical use [83]. Given these issues, liquid biopsy has yet to be included among the guideline-based diagnostic pathways. Accordingly, at present, this technique should be viewed as a strictly regulated research tool, the clinical application of which requires further validation based on the findings of multi-center prospective studies.
In recent years, with respect to the diagnostic evaluation of pancreatic diseases, there has been substantial progress in the evolution and application of AI technologies, particularly radiomics, ML, and deep learning, providing innovative, non-invasive tools for clinical decision-making. Radiomics enables the high-throughput extraction of quantitative features (e.g., texture, intensity, and wavelets) from CT, MRI, and PET-CT images, which are subsequently analyzed using advanced computational methods [84]. This approach has proven to be highly effective in differentiating AIP from PC. Park et al. [85], for example, developed a CT-based radiomic nomogram that achieved 95.2% accuracy in distinguishing PDAC from AIP. Similarly, it has been reported that a dual-phase 18F-FDG PET-CT radiomic ML model achieved a diagnostic accuracy of 89.9% in differentiating PC from AIP [86].
Deep learning, a subset of AI, employs convolutional neural networks (CNNs) to
automatically extract hierarchical image features, and has been demonstrated to
consistently outperform conventional ML techniques. For example, Liu et
al. [87] developed a CT-based CNN model that can be used to accurately
distinguish PC from non-neoplastic pancreatic tissue, showing excellent
generalizability among multi-center and multi-ethnic datasets, with diagnostic
accuracies and sensitivities surpassing those of radiologists, particularly in
detecting early tumors measuring
More recently, multi-modal AI strategies have emerged that integrate radiological imaging, biochemical markers, clinical information, and even histopathology into unified diagnostic frameworks. These approaches contribute to addressing the inherent limitations of single modalities and provide more robust and individualized diagnostic solutions. In this regard, Cui et al. [89] developed a multi-modal AI model that combines EUS imaging with clinical variables, yielding superior performance in differentiating solid pancreatic lesions. When used to assess internal and external test datasets, the joint AI model achieved AUCs of 0.996 and 0.955–0.976, respectively, in distinguishing carcinomas from noncancerous lesions. In a randomized crossover trial, AI-assistance was found to contribute to significant improvements in the diagnostic accuracy of novice endoscopists from 69% to 90% and sensitivity from 61% to 91% [89]. Representative AI studies for differentiating pancreatic lesions are summarized in Table 2 (Ref. [85, 86, 87, 88, 89]).
| Reference | Sample size | Data source | Algorithms | Study type | Key findings (Performance) |
| Park et al. 2020 [85] | 182 (89 AIP, 93 PDAC) | CT | RF | Retrospective, single-center | AUC: 0.975; Acc: 95.2%; Sens: 89.7%; Spec: 100% (Thin-slice venous phase) |
| Liu et al. 2020 [87] | 690 (370 Ca, 320 Non-Ca) | CT | CNN | Retrospective, multi-center | Internal test: AUC 0.997–0.999, Sens 98.3%; External test: AUC 0.920. Outperformed radiologists in sensitivity (98.3% vs. 92.9%) |
| Liu et al. 2021 [86] | 112 (48 AIP, 64 PDAC) | PET-CT | SVM | Retrospective, single-center | AUC: 0.967; Acc: 89.9%; Sens: 85.3%; Spec: 96.0% |
| Kuwahara et al. 2023 [88] | 933 (22,000 images) | EUS | CNN | Retrospective, single-center | Ca vs. Non-Ca (Test): AUC 0.90, Sens 94%; AIP (Test): Sens 73% |
| Cui et al. 2024 [89] | 758 (447 Ca, 311 Non-Ca) | EUS and Clinical Data | CNN | Randomized crossover trial | Internal test: AUC 0.996; External tests: AUC 0.955–0.976. AI-assistance improved novice endoscopist accuracy from 69% to 90% |
Abbreviations: PDAC, pancreatic ductal adenocarcinoma; Ca, carcinoma; Non-Ca, Non-Carcinoma; Acc, accuracy; Sens, sensitivity; Spec, specificity; AUC, area under the curve; RF, Random Forest; CNN, convolutional neural network; SVM, Support Vector Machine; CT, computed tomography; EUS, endoscopic ultrasound; PET-CT, positron emission tomography-computed tomography; AIP, autoimmune pancreatitis.
However, despite this impressive progress, several challenges have limited the widespread clinical application of AI. In addition to the well-established “black-box” problem, most published models developed for pancreatic lesions are based on the use of single-center, retrospective datasets. Moreover, the training and deployment of these models necessitate the use of substantial computational resources and continuous information technology support. In addition, integrating AI software into existing image archiving and communication and hospital information systems is technically complex and often dependent on specific vendors [90, 91, 92], whereas clinicians require dedicated AI training, which currently remains limited and is primarily confined to academic settings [93, 94, 95].
In addition to technical and logistical issues, ethical and legal considerations remain unresolved. Large-scale model training is reliant on access to imaging data, clinical records, and sometimes genomic data, the use of all of which must comply with strict data protection regulations. Even after de-identification, multi-modal data integration may pose a risk of re-identification. In addition, the responsibility for AI-related diagnostic errors is unclear, and the current medicolegal and regulatory frameworks offer limited guidance on allocating liabilities among software developers, healthcare institutions, and individual clinicians [96, 97].
Overall, AI technologies hold considerable promise for improving the differential diagnosis of AIP and PC, although at the current stage of development, these should be viewed as decision-support tools. Future work should focus on explainable AI, large-scale multi-center validation, and integration of AI into multidisciplinary team workflows to support routine clinical use.
In clinical practice, the accurate differentiation between AIP and PC is essential for guiding treatment and improving prognosis. According to the National Comprehensive Cancer Network (NCCN) guidelines for Pancreatic Adenocarcinoma (Version 2.2025) [98], patients with suspected PC should be managed by multidisciplinary teams in specialized centers, whereas the ICDC and European guidelines on IgG4-related digestive disease emphasize a stepwise, multi-modal approach integrating pancreatic imaging, serology, other organ involvement, histology, and steroid response [5, 6]. On this basis, the diagnostic workup can be organized into three stages, namely initial screening, advanced evaluation, and final validation.
Initial screening is dependent on non-invasive methods, including the use of serological markers (IgG4 and CA19-9) and cross-sectional imaging (contrast-enhanced CT or MRI with MRCP). The aim of this stage is to identify the features characteristic of AIP or suggestive of PC, thereby facilitating a stratification of patients into clear diagnostic categories or indeterminate groups requiring further investigation.
For cases with inconclusive or atypical findings following an initial screening, advanced evaluation is warranted, with a focus on EUS-FNB/FNA or ERCP to obtain histological or cytological confirmation. Assessment of extra-pancreatic organ involvement is equally important, with symmetrical inflammatory involvement of the bile ducts, salivary glands, or kidneys being indicative of AIP, whereas PC often presents with metastatic disease. By delineating metabolic patterns, PET-CT can provide complementary information. It is important to note that at the present stage of development, advanced techniques, such as liquid biopsy and AI/radiomics models, should be regarded as research-stage adjuncts. Although their application may be considered in select cases (e.g., in cases for which tissue sampling is non-diagnostic or contraindicated), they should not replace standard histological diagnosis or guideline-based imaging interpretation.
The final validation should be performed under the supervision of a multidisciplinary team. In cases with a strong clinical and imaging suspicion of AIP but insufficient pathological evidence, a short-term steroid trial should be cautiously undertaken, provided that malignancy is rigorously excluded by high-quality imaging and, whenever feasible, EUS-guided tissue sampling. Whereas symptomatic and radiological improvement within 2–4 weeks would be strongly indicative of AIP, a lack of response raises the suspicion of PC. However, given the risk of masking tumor-related inflammation and delaying curative surgery or systemic therapy, such diagnostic steroid trials should be avoided in cases of patients with a high clinical suspicion of PC. A pragmatic three-stage diagnostic algorithm (initial screening, advanced evaluation, and final validation) is presented in Fig. 1.
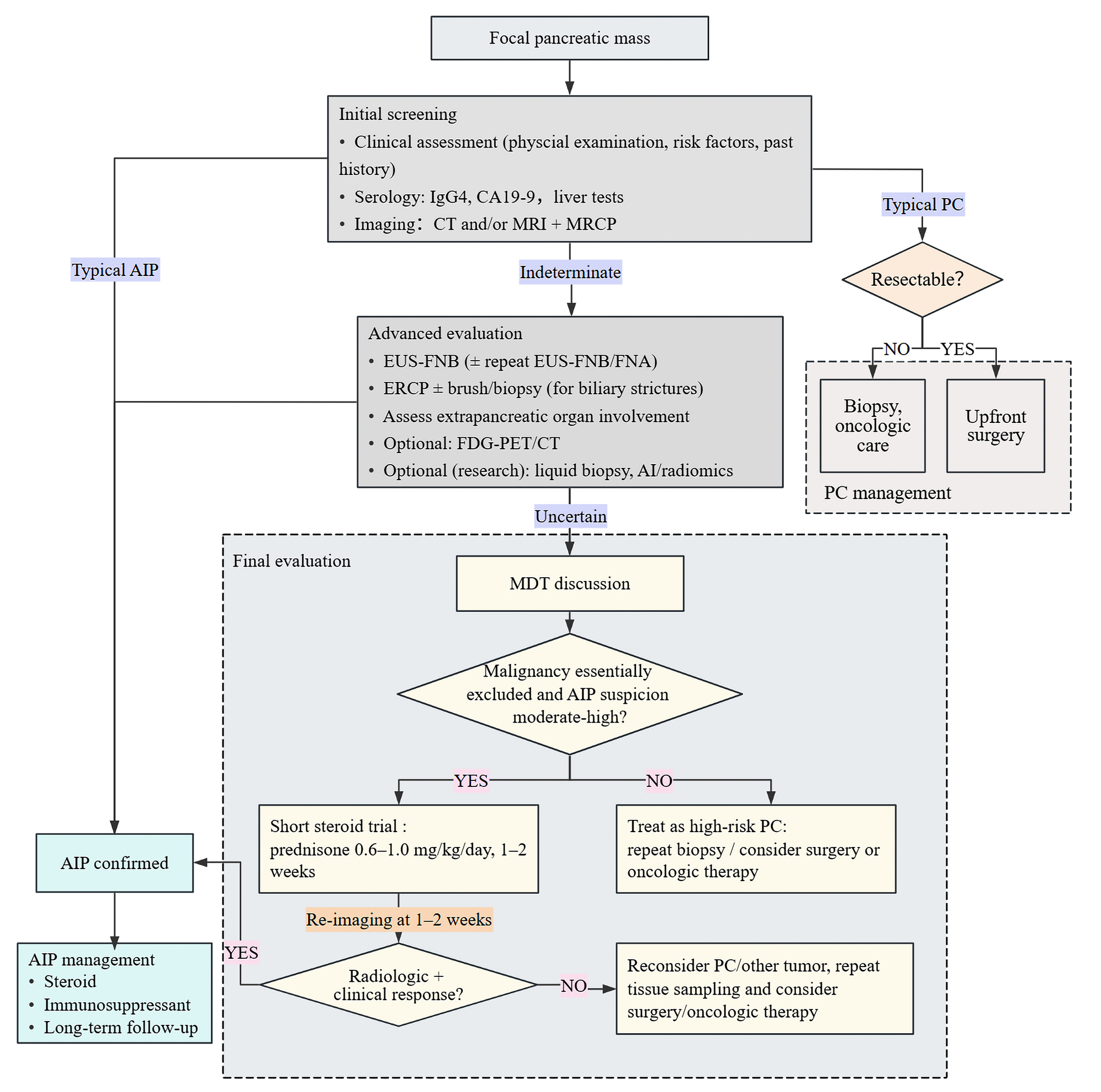 Fig. 1.
Fig. 1.
Proposed diagnostic workflow for differentiating autoimmune pancreatitis from pancreatic cancer. The figure was created using ProcessOn (https://www.processon.com/). The authors have no financial or personal relationship with ProcessOn, and the use of this tool does not imply any endorsement. CT, computed tomography; MRI, magnetic resonance imaging; MRCP, magnetic resonance cholangiopancreatography; AIP, autoimmune pancreatitis; PC, pancreatic cancer; EUS, endoscopic ultrasound; FNA, fine-needle aspiration; FNB, fine-needle biopsy; ERCP, endoscopic retrograde cholangiopancreatography; FDG PET-CT, fluorodeoxyglucose positron emission tomography computed tomography; MDT, multidisciplinary team; CA19-9, carbohydrate antigen 19-9.
Surgical intervention should be reserved for patients with suspected resectable PC or when malignancy cannot be excluded after comprehensive evaluation. Patients who are unresponsive to steroid therapy, or who have a recurrent disease, should also undergo multidisciplinary team consultation to assess surgical indications.
Although novel tools such as AI-based imaging analysis and liquid biopsy show promise in complex scenarios, they remain investigational and have yet to be incorporated into major guidelines. When these results conflict with those of the histopathological or multidisciplinary team assessments, clinical decisions should be based on the latter. Further multi-center prospective studies are required to clarify the role of these technologies in diagnostic pathways.
This study has several limitations. First, most available evidence is derived from single-center retrospective studies with relatively small sample sizes and considerable variability in imaging protocols and reference standards, which may limit the generalizability of the findings. Second, the real-world performance and clinical utility of emerging tools, such as liquid biopsy and AI-based diagnostic models, require rigorous external validation. Ongoing clinical trials are evaluating these diagnostic approaches, particularly AI-driven imaging analysis and liquid biopsy, for early detection (Table 3). The findings of these studies are important in informing and refining future diagnostic pathways. Future research should focus on large-scale multi-center prospective studies to confirm these findings and determine how these technologies can be used safely and effectively in routine clinical care.
| NCT number | Primary focus | Study design | Population enrollment | Status |
| NCT07117045 | AI imaging: Low-dose CT screening for pancreatic cancer. | Prospective, Multi-center, Observational | Individuals undergoing low-dose CT | Recruiting |
| NCT06638866 | AI imaging: Opportunistic screening for pancreatic lesions via non-contrast CT. | Prospective, Multi-center, Observational | Individuals undergoing routine CT | Recruiting |
| NCT06824909 | AI pathology: Rapid on-site evaluation of EUS-FNA cytology smears. | Prospective, Multi-center, Observational | Patients with solid pancreatic lesions | Recruiting |
| NCT06753318 | Multimodal AI: Integration of EUS images and clinical data for diagnosing solid lesions. | Prospective, Multi-center, Randomized controlled trial | Patients with solid pancreatic lesions | Not yet recruiting |
| NCT06564571 | AI imaging: Automated detection of pancreatic abnormalities during EUS. | Prospective, Single-Center, Observational | Patients undergoing EUS | Recruiting |
| NCT06283576 | Liquid Biopsy: Multi-omics analysis for early detection of pancreatic cancer. | Prospective, Single-Center, Observational | Early-stage PC, IPMN, high-risk individuals, healthy controls | Enrolling by invitation |
| NCT06108531 | Liquid Biopsy: Exosome-based diagnostic strategy for pancreatic cancer. | Prospective, Single-Center, Observational | Patients with PC, benign pancreatic lesions, and healthy controls. | Enrolling by invitation |
Abbreviations: AI, artificial intelligence; CT, computed tomography; EUS, endoscopic ultrasound; FNA, fine-needle aspiration; IPMN, intraductal papillary mucinous neoplasm; PC, pancreatic cancer.
Note: All information in this table was extracted from http://clinicaltrials.gov/ on 25 November 2025.
In this review, we describe a new three-stage diagnostic algorithm that may assist clinicians in distinguishing AIP from PC. This structured framework brings together conventional and emerging diagnostic tools, and offers clinicians a clear and practical pathway. Using current evidence to develop an applied strategy represents an advance compared with traditional descriptive reviews and has the potential to reduce misdiagnoses and support optimal management. However, prospective validation will be required to consolidate this approach.
• Autoimmune pancreatitis and pancreatic cancer are often characterized by similar symptoms and imaging features, making differentiation difficult and increasing the risk of misdiagnosis.
• In this review, we summarize the current multidisciplinary evidence obtained from the fields of epidemiology, serology, imaging, and histopathology, and evaluate the potential application of emerging tools such as liquid biopsy and artificial intelligence.
• We propose a practical three-stage diagnostic algorithm that may guide clinicians in combining conventional and advanced diagnostic methods.
• Accurate diagnosis requires a multidisciplinary approach, including serology, cross-sectional imaging, endoscopic ultrasound-guided tissue sampling, and in selected cases, cautiously assessed steroid trials.
Not applicable.
QW: Conceptualization, Literature review, Data curation, Investigation, Writing—original draft preparation and revision. BW: Conceptualization, Supervision, Funding acquisition, Writing—review and editing. Both authors contributed to the important editorial changes in the manuscript. Both authors read and approved the final manuscript. Both authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Not applicable.
Not applicable.
The study was supported by Tongji Hospital Scientific Research Fund Project (No. 2023A14 to BW).
The authors declare no conflict of interest.
References
Publisher’s Note: IMR Press stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

