1 Department of Obstetrics, Suzhou Affiliated Hospital of Nanjing Medical University, Suzhou Municipal Hospital, 215000 Suzhou, Jiangsu, China
†These authors contributed equally.
Academic Editor: Shigeki Matsubara
Abstract
Background: To
evaluate the efficacy and safety of
carbetocin compared with oxytocin for prevention of
postpartum hemorrhage (PPH) after elective
cesarean section in Chinese high risk women. Methods: This was a
single-center, prospective, randomized, open-label, controlled trial recruiting
852 pregnant women with one or more PPH risk factors between
April 2017 and August 2019. Pregnant woman who was scheduled for an elective
cesarean section were randomly assigned to receive carbetocin or oxytocin for
prevention of PPH. The primary efficacy endpoint was the proportion of additional
uterotonics. Results: A total of 852 pregnant women were
randomly assigned to receive carbetocin (n = 442) or oxytocin (n = 410). The
baseline characteristics were comparable between the two groups.
The carbetocin group had lower proportion of requiring
additional uterotonics (18.4% vs. 24.4%, p = 0.03 in full analysis set
[FAS] analysis) to the oxytocin group. The amount of blood loss (intrapartum or
postpartum) was no statistically significant difference (all p
Keywords
- Carbetocin
- Cesarean section
- Postpartum hemorrhage
- Oxytocin
Postpartum hemorrhage (PPH) constitutes a major cause of maternal mortality and postpartum hysterectomy [1]. It is estimated that 140,000 women worldwide die of PPH every year [2]. The most frequent cause of PPH is uterine atony, resulting in up to 80% of PPH [3]. It is recommended to systematically use uterotonics immediately after delivering the newborn for prevention of PPH [4].
Oxytocin is the most widely used uterotonic agent with a rapid
onset of action and a good safety profile [5]. However, due to its short
half-life (4–10 minutes), continuous or frequent repeated administration is
required [6]. Carbetocin, a long-acting synthetic oxytocin analogue, has a longer
duration of action than oxytocin (half-life 85–100 minutes) [7].
The structural difference with oxytocin makes carbetocin more
stable, thereby avoiding the cleavage of aminopeptidase and disulfide compounds
[8]. Compared with oxytocin, carbetocin has been shown to reduce blood loss and
the need for additional uterotonics, and decrease the risk of PPH in cesarean
section [9, 10, 11]. Cesarean section is a well-known risk factor for PPH [6]. The
caesarean section rate in China is about 36.7% in 2018, which is higher than the
World Health Organization recommended proportion of 15% [12]. In our clinical
practice, we usually encounter the PPH risk maternal population (such as scarred
uterus, breech position, uterine fibroid or
This study was a single-center, prospective, randomized, controlled trial comparing Carbetocin and Oxytocin in prevention of PPH after elective cesarean section. A total of 852 pregnant women with one or more PPH risk factors scheduled for an elective cesarean section were recruited between April 2017 and August 2019. This trial was registered with Chinese Clinical Trial Registry (ChiCTR1800015613), and conducted in accordance with Declaration of Helsinki and Good Clinical Practice Guidelines. The protocol had been approved by ethics committee institutional review board of participating center (K2016062), and written informed consent was obtained from each participant.
Women with one or more PPH risk factors, scheduled for cesarean section on term
pregnancy were eligible to enter this study. The indications for elective
cesarean section and PPH risk factors included scarred uterus (the uterus of a
woman that had undergone myomectomy or previous cesarean section), uterine
fibroid, breech position, and age
All eligible subjects were randomly assigned to carbetocin group or oxytocin group by a computer-generated coding system (Fig. 1). All women underwent low transverse cesarean section under spinal anesthesia, which performed by an obstetrician team including senior physicians and anesthesiologists led by a department chief to ensure the quality and safety. All doctors in our hospital had undergone strict training and assessment which were similar in the level of expertise. The anesthetic drugs included butorphanol (1 mg, intraspinal injection; H20143106, Jiangsu Hengrui Pharmaceutical Co., Ltd., Lianyungang, China), esmolol (1 g, intravenous injection; H19991058, Qilu Pharmaceutical Co., Ltd., Jinan, China), ropivacaine (20 mg, intravenous injection; H20100103, Naropin, AstraZeneca Pharmaceuticals Ltd., Shanghai, China), flurbiprofen axetil (50 mg, intravenous injection; H20041508, Beijing Taide Pharmaceutical Co. Ltd., Beijing, China) and granisetron hydrochloride (20 mg, intravenous injection; H10970243, Ningbo Tianheng Pharmaceutical Co., Ltd., Ningbo, China). In addition, cefoxitin sodium (2.0 g, intravenous drip; H20057973, Yangzijiang Pharmaceutical Group Co., Ltd., Taizhou, China) was used for infection prophylaxis.
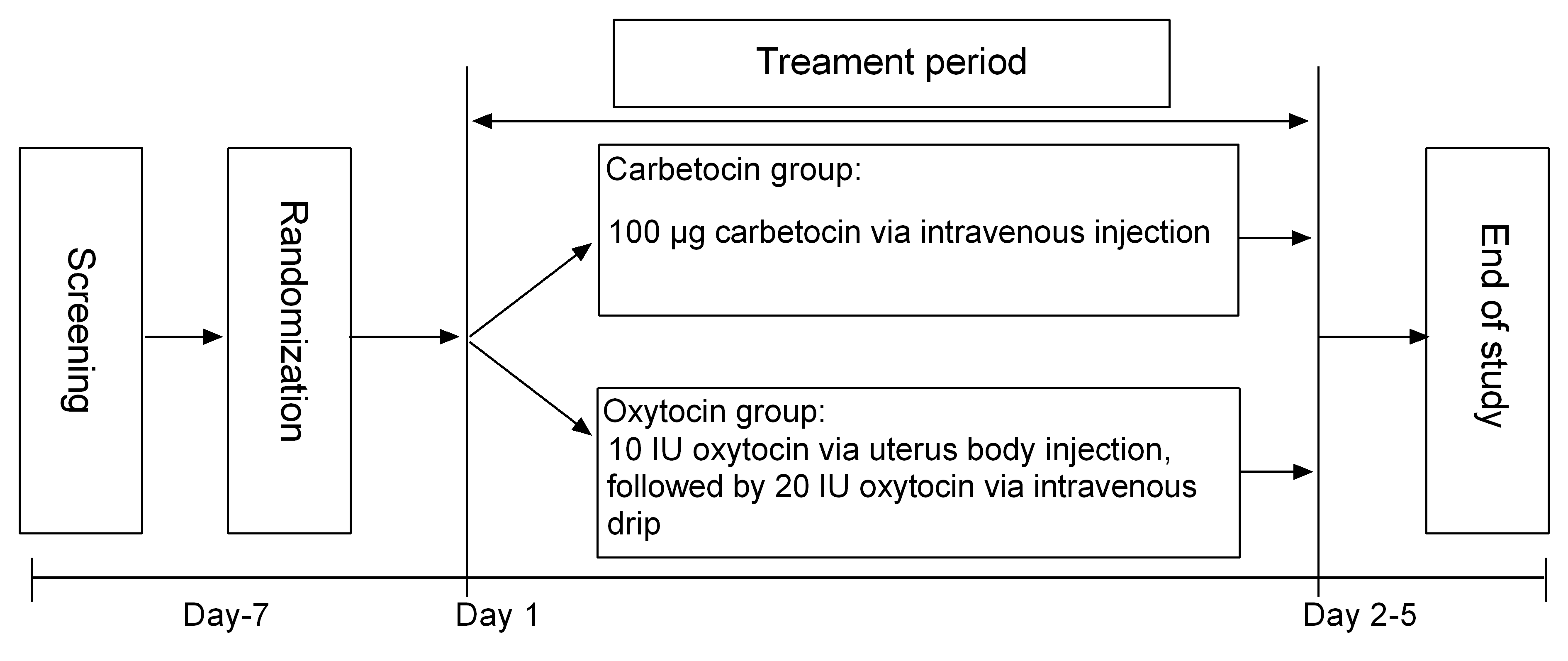 Fig. 1.
Fig. 1.Study design.
After delivering the newborn, women in the carbetocin group received
100
The baseline characteristics, additional uterotonics usage, blood loss during surgery, PPH, antepartum hemoglobin and postpartum hemoglobin, maternal and neonatal outcomes of patients were recorded and analyzed. Blood loss was calculated by measuring the volume in the suction bottle and the absorption in the surgical drapes, gauzes, and pads.
The primary efficacy endpoint was the proportion of women requiring additional
uterotonics after administration of the investigative drug
(carbetocin or oxytocin). The primary efficacy endpoint was chosen instead of the
occurrence of PPH for the following reasons: firstly, the amount of bleeding was
known to be underestimated [13]; secondly, continuous and slow
bleeding from the surgical incision contributed to the incidence and severity of
PPH [14]; lastly, the administration of several additional uterotonics may be
used to successfully avoid bleeding in some cases.
Secondary efficacy endpoints included the
following: (1) the amount of intrapartum blood loss; (2) the incidence of blood
loss
The safety was evaluated by the maternal and neonatal outcomes. The organ damage (heart, lung, brain, liver or kidney), need for respirator support and hemofiltration or plasmapheresis were used to evaluate the maternal outcomes. Meanwhile, Apgar score and neonatal intensive care unit (NICU) admission were selected to evaluate the neonatal outcomes.
The sample size calculation for this trial was based on primary endpoint
according to the parameter of previous study [15]. The investigator anticipated a
better performance of carbetocin on the efficacy at a significant level of
Efficacy analysis was performed based on the intention-to-treat population. All
results were analyzed in full analysis set (FAS) and per-protocol set (PPS).
SPSS version 22.0 (SPSS Institute, Chicago, IL, USA) was used
to perform the statistical analyses. Quantitative data were expressed as means
852 eligible pregnant women were randomly assigned to receive carbetocin (n = 442) or oxytocin (n = 410). A total of 841 women were included in FAS analysis (carbetocin group: n = 440; oxytocin group: n = 401). The reasons for not including in FAS analysis was a need for emergency surgery during night shift (n = 11). 827 patients were included in the PPS analysis (carbetocin group: n = 439; oxytocin group: n = 388). 14 patients who were treated with oxytocin mistakenly due to PPH were excluded from PPS analysis (Fig. 2). Baseline characteristics of patients were shown in Table 1. The two groups were generally well balanced for the maternal age, body mass index (BMI), gestational age, parity, antepartum hemoglobin, antepartum hematocrit, pregnancy complications, birth weight and PPH risk factors.
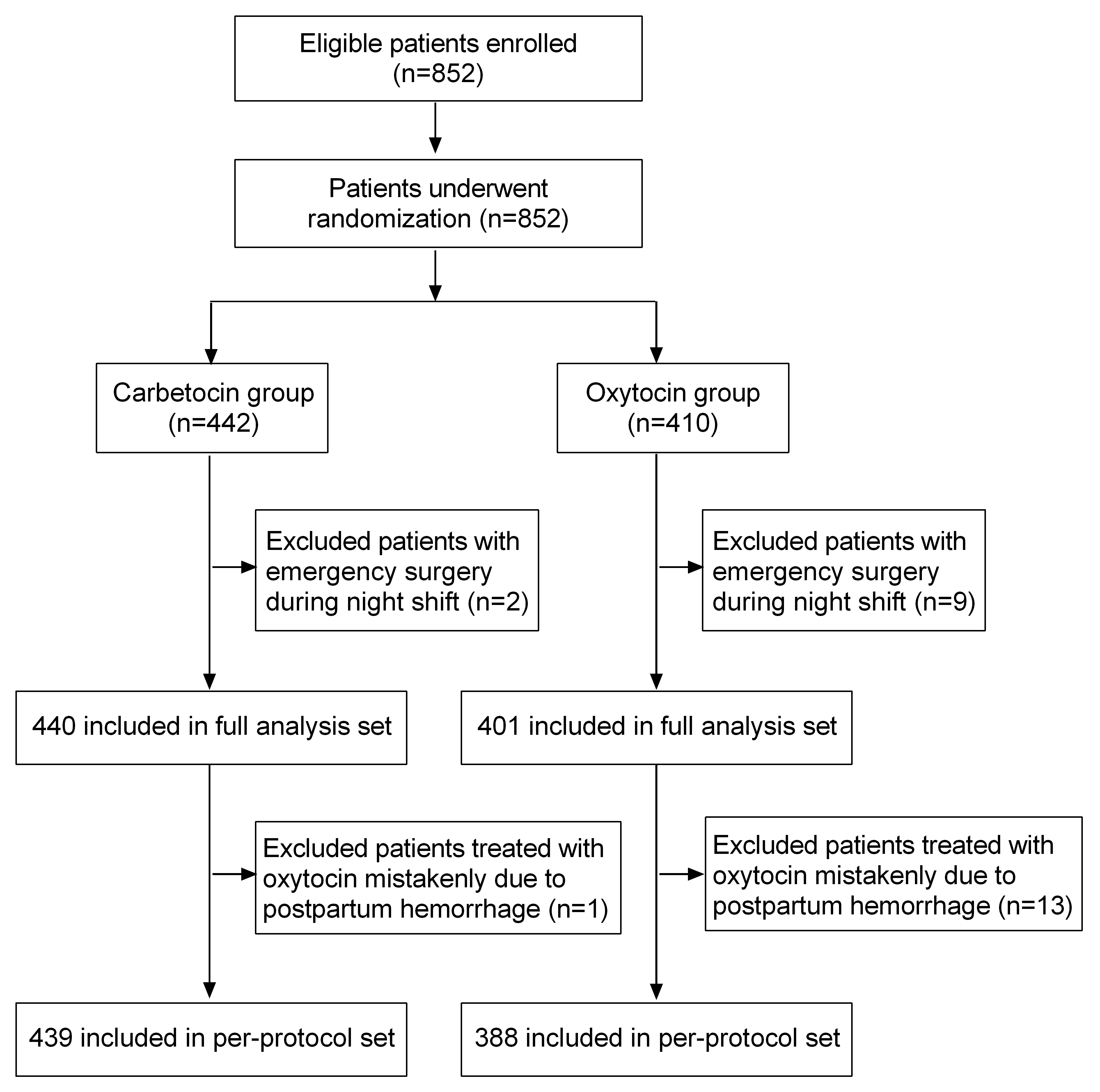 Fig. 2.
Fig. 2.Patients flowchart.
| Characteristic | FAS analysis | PPS analysis | |||||
| Carbetocin group (n = 440) | Oxytocin group (n = 401) | p value | Carbetocin group n = 439) | Oxytocin group (n = 388) | p value | ||
| Maternal age (year, Mean |
31.6 |
31.7 |
0.08 | 31.6 |
31.7 |
0.06 | |
| BMI (kg/m |
27.6 |
27.8 |
0.4 | 27.6 |
27.8 |
0.4 | |
| Gestational age (weeks, Mean |
39.1 |
39.2 |
0.9 | 39.1 |
39.2 |
0.9 | |
| Parity (n, %) | 0.8 | 0.6 | |||||
| Nulliparous | 107 (24.3) | 94 (23.4) | 107 (24.4) | 88 (22.7) | |||
| Multiparous | 333 (75.7) | 307 (76.6) | 332 (75.6) | 300 (77.3) | |||
| Antepartum hemoglobin (g/L, Mean |
116.6 |
117.4 |
0.3 | 116.6 |
117.2 |
0.3 | |
| Normal ( |
324 (73.6) | 301 (75.1) | 323 (73.6) | 290 (74.7) | |||
| Mild anemia (100–110 g/L, n/%) | 80 (18.2) | 78 (19.5) | 80 (18.2) | 76 (19.6) | |||
| Moderate anemia (70–100 g/L, n/%) | 36 (8.2) | 22 (5.5) | 36 (8.2) | 22 (5.7) | |||
| Severe anemia ( |
0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |||
| Antepartum hematocrit (Mean |
0.3 |
0.4 |
0.1 | 0.3 |
0.4 |
0.1 | |
| Hypertension (n, %) | 6 (1.4) | 1 (0.2) | 0.2 | 6 (1.4) | 1 (0.3) | 0.2 | |
| Uterine fibroids (n, %) | 6 (1.4) | 13 (3.2) | 0.1 | 6 (1.4) | 13 (3.4) | 0.1 | |
| Diabetes (n, %) | 94 (21.4) | 81 (20.2) | 0.7 | 94 (21.4) | 79 (20.4) | 0.7 | |
| Birth weight (g, Mean |
3432.6 |
3400.4 |
0.25 | 3431.4 |
3999.9 |
0.7 | |
| PPH risk factors (n, %) | 0.2 | 0.8 | |||||
| Scarred uterus | 319 (72.7) | 294 (78.6) | 318 (72.6) | 287 (74.0) | |||
| Uterine fibroid | 2 (0.5) | 1 (0.2) | 2 (0.5) | 1 (0.3) | |||
| Breech position | 85 (19.4) | 70 (14.0) | 85 (19.4) | 67 (17.3) | |||
| Age |
19 (4.3) | 17 (4.2) | 19 (4.3) | 16 (4.1) | |||
| FAS, full analysis set; PPS, per-protocol set; BMI, body mass index; SD, standard deviations; PPH, postpartum hemorrhage. | |||||||
The primary and secondary efficacy endpoints were analyzed in FAS and PPS. When the conclusions of the two were inconsistent, the FAS analysis results were mainly considered.
The primary efficacy endpoint of the use of additional uterotonics (FAS analysis) occurred in 81 (18.4%) cases in the carbetocin group, as compared with 98 (24.4%) in the oxytocin group (Table 2). Compared with oxytocin, carbetocin showed the superiority in terms of the primary efficacy endpoint (FAS analysis, p = 0.03, odds ratio = 0.7, 95% CI 0.5–0.9).
The amount of intrapartum blood loss was 370.3
| Outcomes | FAS analysis | PPS analysis | |||||
| Carbetocin group (n = 440) | Oxytocin group (n = 401) | p value | Carbetocin group (n = 439) | Oxytocin group (n = 388) | p value | ||
| Primary efficacy endpoint | |||||||
| The use of additional uterotonics (n, %) | 81 (18.4) | 98 (24.4) | 0.03 | 80 (18.2) | 89 (22.9) | 0.1 | |
| Secondary efficacy endpoint | |||||||
| Intrapartum blood loss (mL, Mean |
370.3 |
386.6 |
0.2 | 370.3 |
384.7 |
0.3 | |
| Blood loss |
16 (3.6) | 19 (4.7) | 0.4 | 13 (3.0) | 12 (3.1) | 0.9 | |
| Blood loss within 2 hours after delivery (mL, Mean |
421.8 |
456.1 |
0.1 | 421.8 |
456.8 |
0.1 | |
| Blood loss within 24 hours after delivery (mL, Mean |
501.7 |
513.7 |
0.5 | 501.7 |
513.6 |
0.5 | |
| Blood loss |
14 (3.2) | 21 (5.2) | 0.1 | 14 (3.2) | 21 (5.4) | 0.1 | |
| Blood loss |
8 (1.8) | 10 (2.5) | 0.5 | 8 (1.8) | 10 (2.6) | 0.5 | |
| Postpartum hemoglobin (g/L, Mean |
116.7 |
116.8 |
0.9 | 116.7 |
116.9 |
0.8 | |
| Normal ( |
315 (72.1) | 285 (71.4) | 315 (72.1) | 278 (71.7) | |||
| Mild anemia (100–110 g/L, n/%) | 77 (17.6) | 84 (21.1) | 77 (17.6) | 80 (20.6) | |||
| Moderate anemia (70–100 g/L, n/%) | 45 (10.3) | 30 (7.5) | 45 (10.3) | 30 (7.7) | |||
| Severe anemia ( |
0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | |||
| Hemostatics (n, %) | 1 (0.2) | 5 (1.2) | 0.2 | 1 (0.2) | 4 (1.0) | 0.3 | |
| Blood transfusion (n, %) | 1 (0.2) | 6 (1.5) | 0.1 | 1 (0.2) | 6 (1.6) | 0.1 | |
| Additional surgical interventions (n, %) | 26 (5.9) | 20 (5.0) | 0.6 | 26 (5.9) | 19 (4.9) | 0.5 | |
| Uterine massage (n, %) | 1 (0.2) | 4 (1.0) | 0.3 | 1 (0.2) | 4 (1.0) | 0.3 | |
| FAS, full analysis set; PPS, per-protocol set; SD, standard deviations. | |||||||
The maternal and neonatal outcomes were listed in Table 3. Regarding maternal outcomes, no poor outcomes such as organ damage (heart, lung, brain, liver or kidney), need for respirator support and hemofiltration or plasmapheresis were observed in carbetocin and oxytocin groups. For neonatal outcomes, mild asphyxia (Apgar score 4–7) was occurred in 9 (2.1%) cases in the carbetocin group, as compared with 5 (1.3%) cases in the oxytocin group. No other poor neonatal outcomes such as severe asphyxia (Apgar score 1–3) or NICU admission were observed in two groups.
| Outcomes | Carbetocin group (n = 439) | Oxytocin group (n = 388) | ||
| Maternal outcomes (n, %) | ||||
| Organ damage | 0 (0.0) | 0 (0.0) | ||
| Need for respirator support | 0 (0.0) | 0 (0.0) | ||
| hemofiltration or plasmapheresis | 0 (0.0) | 0 (0.0) | ||
| Neonatal outcomes (n, %) | ||||
| Apgar score | ||||
| 8–10 | 430 (98.0) | 383 (98.7) | ||
| 4–7 | 9 (2.1) | 5 (1.3) | ||
| 1–3 | 0 (0.0) | 0 (0.0) | ||
| NICU admission | 0 (0.0) | 0 (0.0) | ||
| NICU, neonatal intensive care unit. | ||||
Our study compared the efficacy and safety of initial use of carbetocin or oxytocin for prevention of PPH after elective cesarean section in Chinese high risk women. The present study found that carbetocin significantly reduced the proportion of additional uterotonics after elective cesarean section compared with oxytocin. However, no significant difference was found in the other clinical indicators of blood loss between the two groups, such as postpartum blood loss, postpartum hemoglobin, hemostatics, blood transfusion, additional surgical interventions or uterine massage.
Caesarean section is a risk factor for PPH [16, 17]. Preventive use of uterotonics can reduce mean blood loss, thereby reducing maternal morbidity and mortality [6]. Although oxytocin is the first-line agent in the prevention of postpartum hemorrhage, carbetocin has found its place in modern obstetrics [18]. So far, the best products to prevent PPH have yet to be determined. In present study, we found that the carbetocin group had lower proportion of requiring additional uterotonics (18.4% vs. 24.4%, p = 0.03 in FAS analysis) to the oxytocin group. Similarly, Attilakos et al. [19] found a reduced number of additional uterotonic agents were requried in the carbetocin group compared with oxytocin group. Another study by Dansereau et al. [20] also concluded that carbetocin compared with oxytocin resulted in a less use of further uterotonic agents. These results demonstrated that carbetocin required lower rate of additional uterotonics than oxytocin for prevention of PPH after elective cesarean section in Chinese high risk women.
A study concluded by Maged et al. [21] among pregnant women who had at
least two PPH risk factors showed that carbetocin was superior to oxytocin
regarding postpartum blood loss, the change in hemoglobin level, and the need for
uterine massage after vaginal delivery. In addition, several previous studies had
showed that carbetocin reduced the blood loss and the risk of PPH in caesarean
section compared to oxytocin [9, 10, 22]. However, similar results were not found
in our research. The present study showed that no significant differences in the
postpartum blood loss, postpartum hemoglobin, hemostatics, blood transfusion,
additional surgical interventions or uterine massage was found between the two
groups. The discrepancy between previous reports and our findings may be result
from the method of calculation of blood loss during caesarean section and the
population involved in this study. In present study, blood loss was calculated by
measuring the volume in the suction bottle and the absorption in the surgical
drapes, gauzes, and pads. The measurement inevitably included a certain amount of
amniotic fluid, making the assessment less accurate. In addition, when a pregnant
woman had a tendency to develop PPH, in order to deal with it in time, the
cut-off value of 800 mL was used in our study to avoid the occurrence of PPH.
Besides, pregnant women with one or more PPH risk factors (scarred uterus,
uterine fibroid, breech position, age
Several notable limitations exist in our study. First of all, our study was conducted at a single institute, which had certain limitations in clinical application. Secondly, the safety was evaluated only by the maternal and neonatal outcomes, and the drugs side effects in this study were ignored. Thirdly, the blood loss could not be measured precisely. This study needs to be repeated in a multicenter setting to further identify the impact of carbetocin on the aforementioned factors and verify our findings and hypothesis.
Carbetocin required lower rate of additional uterotonics than oxytocin for prevention of PPH after elective cesarean section in Chinese high risk women. Carbetocin was comparable to oxytocin in postpartum blood loss, postpartum hemoglobin, hemostatics, blood transfusion, additional surgical interventions or uterine massage.
LPZhu and YW—designed the research study. SYK, LPZhou, YFY and LY—collected data. SYK, LPZhou, YFY and LY—analyzed the data. LPZhu and YW—provided administrative support. SYK, LPZhou, LPZhu and YW—drafted the article. All authors contributed to editorial changes in the manuscript. All—final approval of the manuscript submitted.
The protocol had been approved by ethics committee institutional review board of participating center (K2016062), and written informed consent was obtained from each participant.
The authors express their gratitude to all those who helped them during the writing of this manuscript. Thanks to all the peer reviewers for their opinions and suggestions.
This work was supported by the Suzhou People’s Well-being Project in China [SS201710] and the Suzhou Introduction of Clinical Expert Team Project [SZYJTD201709].
The authors declare no conflict of interest.
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.


